QUESTION IMAGE
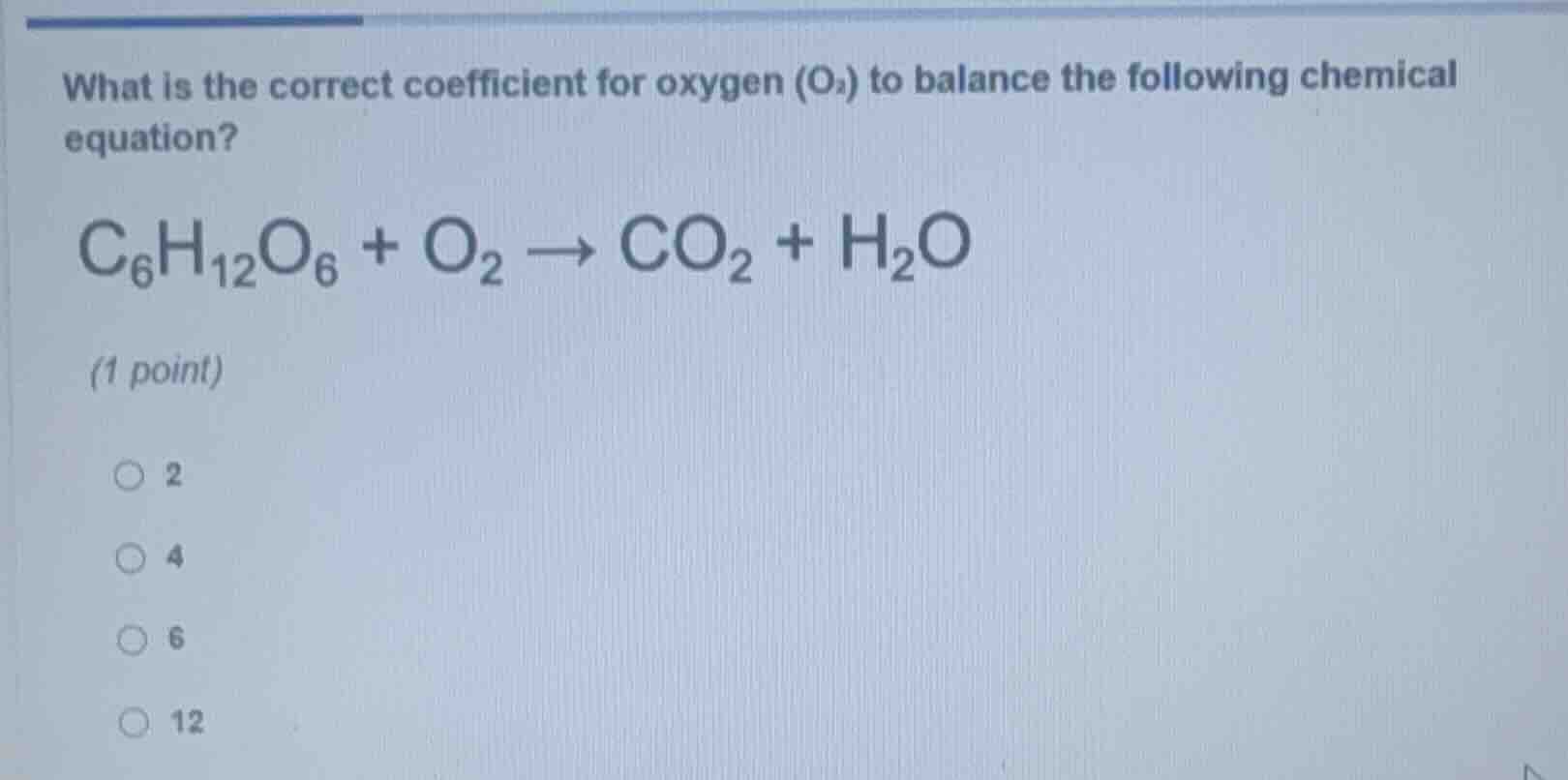
Question
what is the correct coefficient for oxygen (o₂) to balance the following chemical equation?
c₆h₁₂o₆ + o₂ → co₂ + h₂o
(1 point)
○ 2
○ 4
○ 6
○ 12
Step1: Balance Carbon atoms
On the left side, we have 6 C atoms in $\ce{C6H12O6}$. So, we need 6 $\ce{CO2}$ on the right to balance C. So the equation becomes $\ce{C6H12O6 + O2 -> 6CO2 + H2O}$.
Step2: Balance Hydrogen atoms
On the left side, we have 12 H atoms in $\ce{C6H12O6}$. Each $\ce{H2O}$ has 2 H atoms, so we need $\frac{12}{2}=6$ $\ce{H2O}$ molecules. Now the equation is $\ce{C6H12O6 + O2 -> 6CO2 + 6H2O}$.
Step3: Balance Oxygen atoms
Calculate O atoms on the right: In 6 $\ce{CO2}$, we have $6\times2 = 12$ O atoms, and in 6 $\ce{H2O}$, we have $6\times1=6$ O atoms. Total O on the right: $12 + 6=18$. On the left, $\ce{C6H12O6}$ has 6 O atoms. Let the coefficient of $\ce{O2}$ be $x$. So, $6 + 2x=18$. Solving for $x$: $2x=18 - 6=12$, so $x = 6$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6 (corresponding to the option with 6)