QUESTION IMAGE
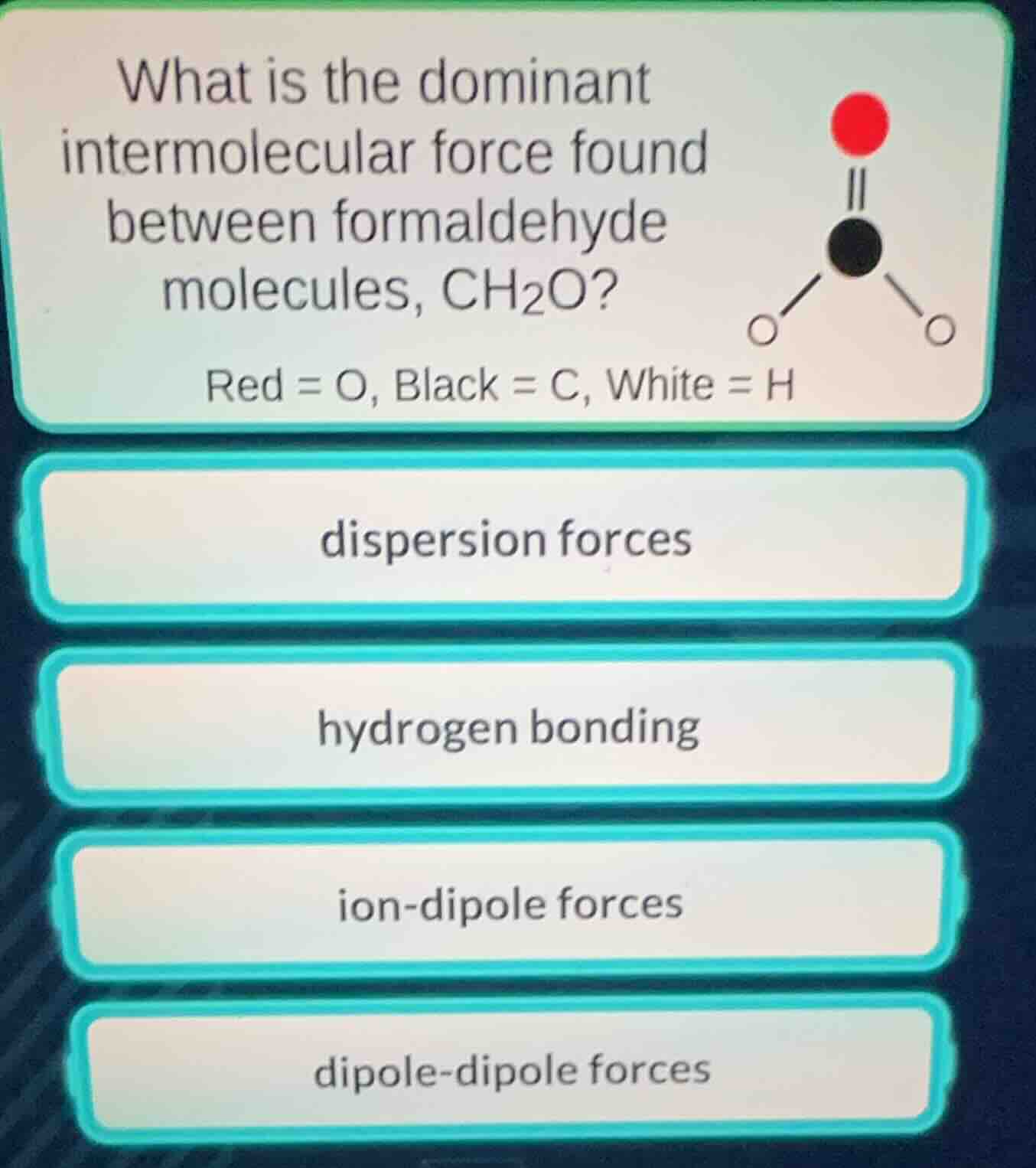
Question
what is the dominant intermolecular force found between formaldehyde molecules, ch₂o?
red = o, black = c, white = h
dispersion forces
hydrogen bonding
ion - dipole forces
dipole - dipole forces
Brief Explanations
- Formaldehyde ($\text{CH}_2\text{O}$) has a trigonal planar geometry with a polar C=O bond, creating a permanent molecular dipole (partial positive on C, partial negative on O).
- Dispersion forces exist between all molecules but are not dominant here because formaldehyde is polar.
- Hydrogen bonding requires a molecule to have an H atom bonded to N, O, or F; formaldehyde has H only bonded to C, so hydrogen bonding is not possible.
- Ion-dipole forces require ions and polar molecules, which are not present between neutral formaldehyde molecules.
- The dominant intermolecular force between polar neutral molecules is dipole-dipole forces, which arise from attractions between the partial charges of adjacent dipoles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
dipole-dipole forces