QUESTION IMAGE
Question
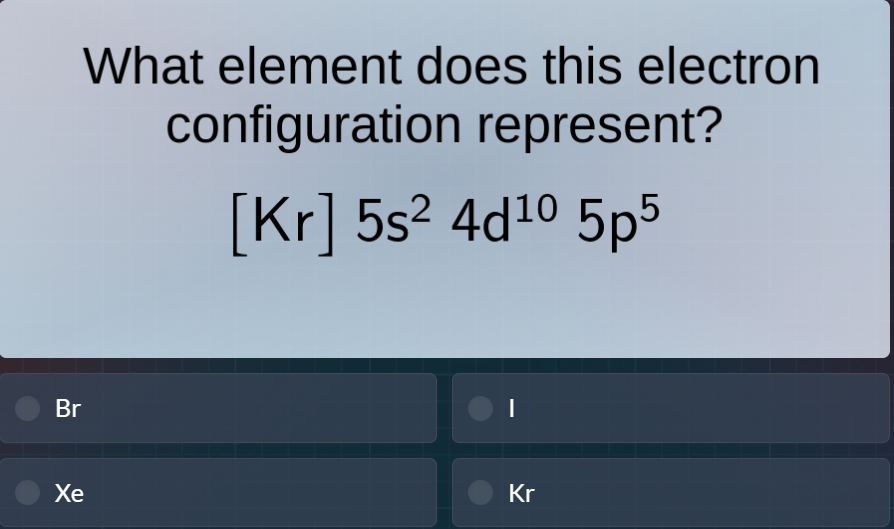
what element does this electron configuration represent?
$kr 5s^2 4d^{10} 5p^5$
br
i
xe
kr
Brief Explanations
- First, find the atomic number of Kr (krypton), which is 36.
- Add the electrons from the valence configuration: $2 + 10 + 5 = 17$.
- Calculate the total atomic number: $36 + 17 = 53$.
- The element with atomic number 53 is iodine (I).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
I. Iodine (I)