QUESTION IMAGE
Question
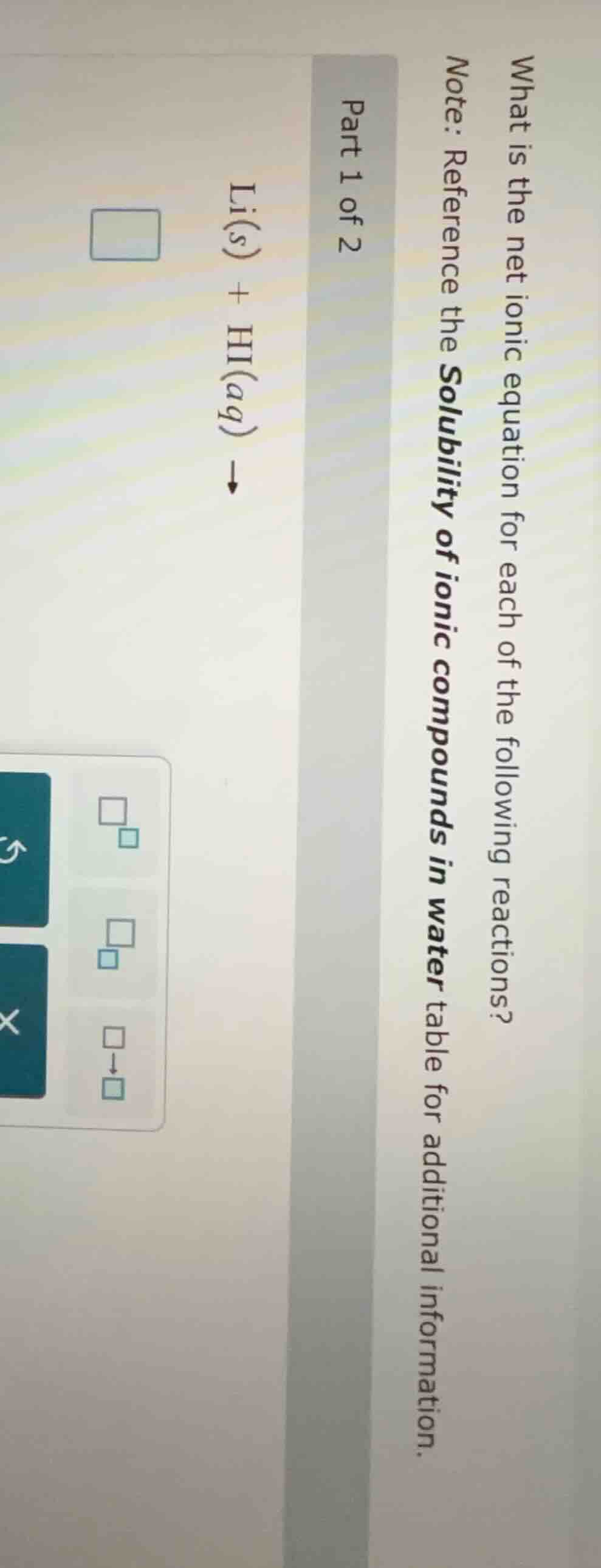
what is the net ionic equation for each of the following reactions? note: reference the solubility of ionic compounds in water table for additional information. part 1 of 2 li(s) + hi(aq) \
ightarrow
Step 1: Identify reactants and their states
The reactants are lithium metal (\( \text{Li}(s) \)) and hydroiodic acid (\( \text{HI}(aq) \)). Hydroiodic acid is a strong acid, so it dissociates completely in water: \( \text{HI}(aq)
ightarrow \text{H}^+(aq) + \text{I}^-(aq) \). Lithium is a solid metal.
Step 2: Determine the products
This is a single - replacement reaction (metal displaces hydrogen from an acid). The products will be lithium iodide (\( \text{LiI} \)) and hydrogen gas (\( \text{H}_2(g) \)). Lithium iodide is soluble in water (since alkali metal salts are soluble), so it dissociates into \( \text{Li}^+(aq) \) and \( \text{I}^-(aq) \).
The overall chemical equation is: \( 2\text{Li}(s) + 2\text{HI}(aq)
ightarrow 2\text{LiI}(aq) + \text{H}_2(g) \)
Step 3: Write the complete ionic equation
Substitute the soluble ionic compounds with their dissociated ions:
\( 2\text{Li}(s) + 2\text{H}^+(aq) + 2\text{I}^-(aq)
ightarrow 2\text{Li}^+(aq) + 2\text{I}^-(aq) + \text{H}_2(g) \)
Step 4: Cancel the spectator ions
The spectator ion here is \( \text{I}^- \) (it appears on both sides of the complete ionic equation). After canceling the spectator ions, we get the net ionic equation.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 2\text{Li}(s) + 2\text{H}^+(aq)
ightarrow 2\text{Li}^+(aq) + \text{H}_2(g) \)