QUESTION IMAGE
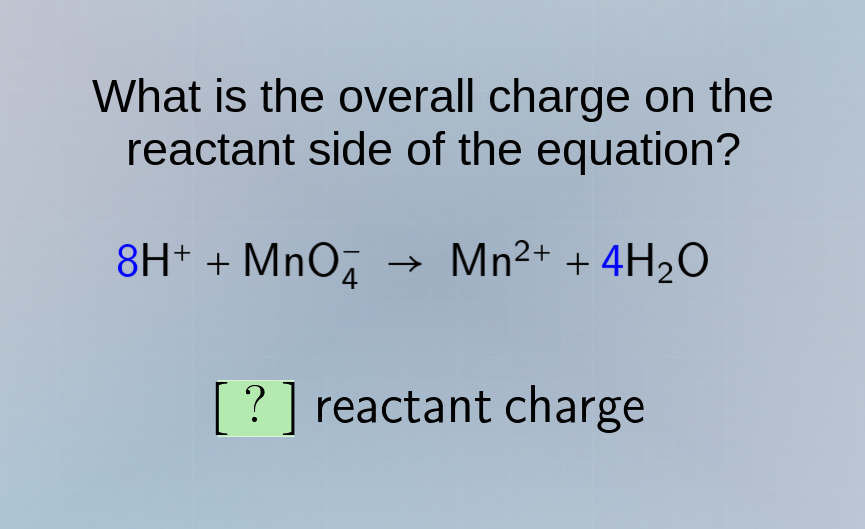
Question
what is the overall charge on the reactant side of the equation?
8h⁺ + mno₄⁻ → mn²⁺ + 4h₂o
? reactant charge
Step1: Calculate charge from \(8\text{H}^+\)
Each \(\text{H}^+\) has a charge of \(+1\), so for \(8\) of them, the total charge is \(8\times(+1) = +8\).
Step2: Add charge from \(\text{MnO}_4^-\)
The \(\text{MnO}_4^-\) ion has a charge of \(-1\). Now, add the charges from \(\text{H}^+\) and \(\text{MnO}_4^-\): \(+8 + (-1)=+7\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(+7\)