QUESTION IMAGE
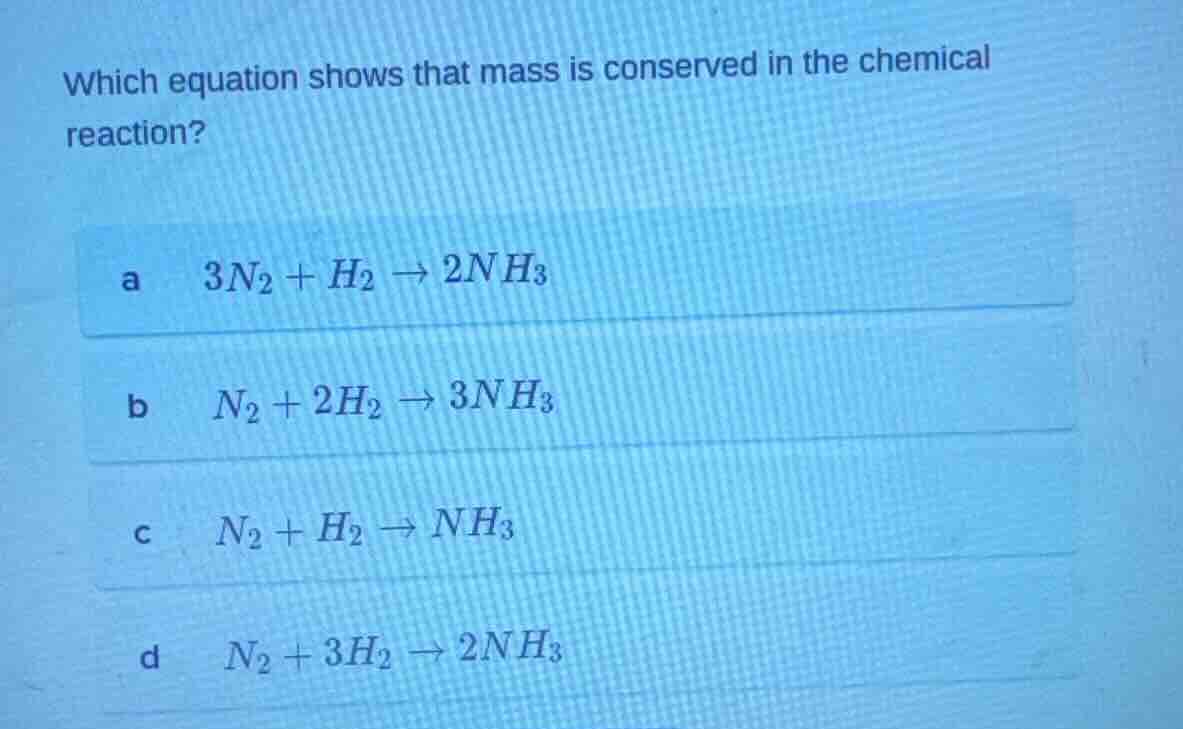
Question
which equation shows that mass is conserved in the chemical reaction?
a $3n_2 + h_2 \
ightarrow 2nh_3$
b $n_2 + 2h_2 \
ightarrow 3nh_3$
c $n_2 + h_2 \
ightarrow nh_3$
d $n_2 + 3h_2 \
ightarrow 2nh_3$
To determine which equation shows mass conservation (balanced chemical equation), we check the number of each atom on both sides.
Step 1: Analyze Option a
Reactants: \(3N_2\) (6 N) and \(H_2\) (2 H). Products: \(2NH_3\) (2 N, 6 H). N atoms not balanced.
Step 2: Analyze Option b
Reactants: \(N_2\) (2 N) and \(2H_2\) (4 H). Products: \(3NH_3\) (3 N, 9 H). N and H not balanced.
Step 3: Analyze Option c
Reactants: \(N_2\) (2 N) and \(H_2\) (2 H). Products: \(NH_3\) (1 N, 3 H). N and H not balanced.
Step 4: Analyze Option d
Reactants: \(N_2\) (2 N) and \(3H_2\) (6 H). Products: \(2NH_3\) (2 N, 6 H). Both N and H are balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. \(N_2 + 3H_2
ightarrow 2NH_3\)