QUESTION IMAGE
Question
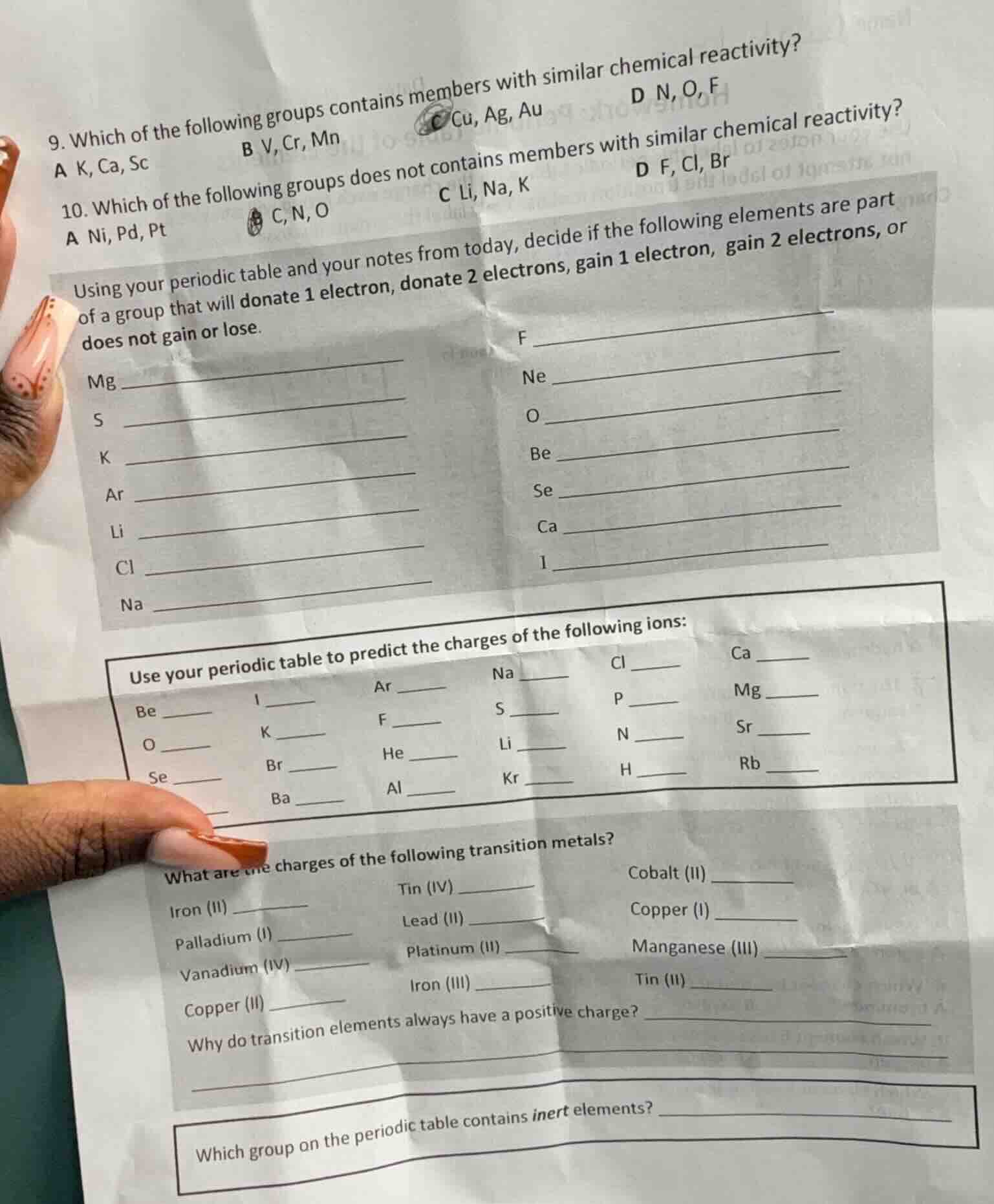
- which of the following groups contains members with similar chemical reactivity?a k, ca, scb v, cr, mnc cu, ag, aud n, o, f10. which of the following groups does not contains members with similar chemical reactivity?a ni, pd, ptc, n, oc li, na, kd f, cl, brusing your periodic table and your notes from today, decide if the following elements are part of a group that will donate 1 electron, donate 2 electrons, gain 1 electron, gain 2 electrons, or does not gain or lose.mg ____s __k __ar __li __cl __na __f __ne __o __be __se __ca __i __use your periodic table to predict the charges of the following ions:be __i __ar __na __cl __ca __o __k __f __s __p __mg __se __br __he __li __n __sr __ba __al __kr __h __rb __what are the charges of the following transition metals?iron (ii) __tin (iv) __cobalt (ii) __palladium (i) __lead (ii) __copper (i) __vanadium (iv) __platinum (iii) __manganese (iii) __copper (ii) __iron (iii) __tin (ii) __why do transition elements always have a positive charge? __which group on the periodic table contains inert elements? ____
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Cu, Ag, Au
B. C, N, O
donate 2 electrons
gain 2 electrons
donate 1 electron
does not gain or lose
donate 1 electron
gain 1 electron
donate 1 electron
gain 1 electron
does not gain or lose
gain 2 electrons
donate 2 electrons
gain 2 electrons
donate 2 electrons
gain 1 electron
+2
-1
0
+1
-1
+2
-2
+1
-1
-2
+3
+2
-2
-1
0
+1
-3
+2
+2
0
+3
+1
+1
+2
+4
+2
+4
+2
+1
+3
+3
+2
+3
+2
Transition elements have valence electrons in d-orbitals and tend to lose these electrons to form positive ions.
Group 18 (Noble Gases)