QUESTION IMAGE
Question
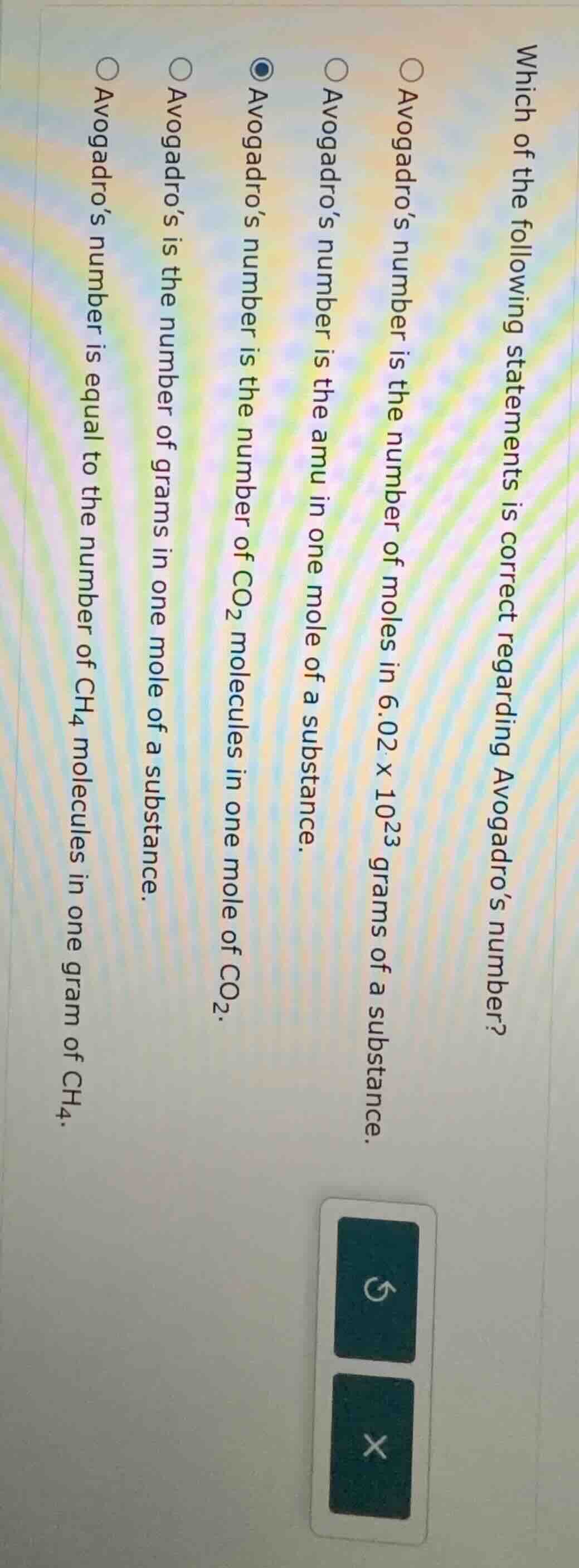
which of the following statements is correct regarding avogadro’s number?
○ avogadro’s number is the number of moles in 6.02 × 10²³ grams of a substance.
○ avogadro’s number is the amu in one mole of a substance.
● avogadro’s number is the number of co₂ molecules in one mole of co₂.
○ avogadro’s is the number of grams in one mole of a substance.
○ avogadro’s number is equal to the number of ch₄ molecules in one gram of ch₄.
- Analyze each option:
- Option 1: Avogadro’s number is the number of particles (atoms, molecules, ions, etc.) in one mole of a substance, not the number of moles in a mass. So this is incorrect.
- Option 2: 1 amu (atomic mass unit) is approximately the mass of one proton or neutron, and the molar mass in grams per mole is numerically equal to the atomic/molecular mass in amu. But Avogadro’s number is about the number of particles in a mole, not amu in a mole. So this is incorrect.
- Option 3: One mole of any substance contains Avogadro’s number of particles (molecules, in the case of $CO_2$). So one mole of $CO_2$ has Avogadro’s number of $CO_2$ molecules. This is correct.
- Option 4: Avogadro’s number is the number of particles in one mole, not the number of grams. The molar mass (in grams) of a substance is equal to its atomic/molecular mass (in amu), but that's different from Avogadro’s number. So this is incorrect.
- Option 5: To find the number of $CH_4$ molecules in 1 gram of $CH_4$, we use molar mass and Avogadro’s number. Molar mass of $CH_4$ is $12 + 4\times1 = 16$ g/mol. Number of moles in 1 gram is $\frac{1}{16}$ mol, and number of molecules is $\frac{1}{16} \times N_A$ (where $N_A$ is Avogadro’s number), which is not equal to $N_A$. So this is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Avogadro’s number is the number of $CO_2$ molecules in one mole of $CO_2$. (Note: Assuming the third option is labeled as C here, based on the standard multiple - choice labeling and the content of the option)