QUESTION IMAGE
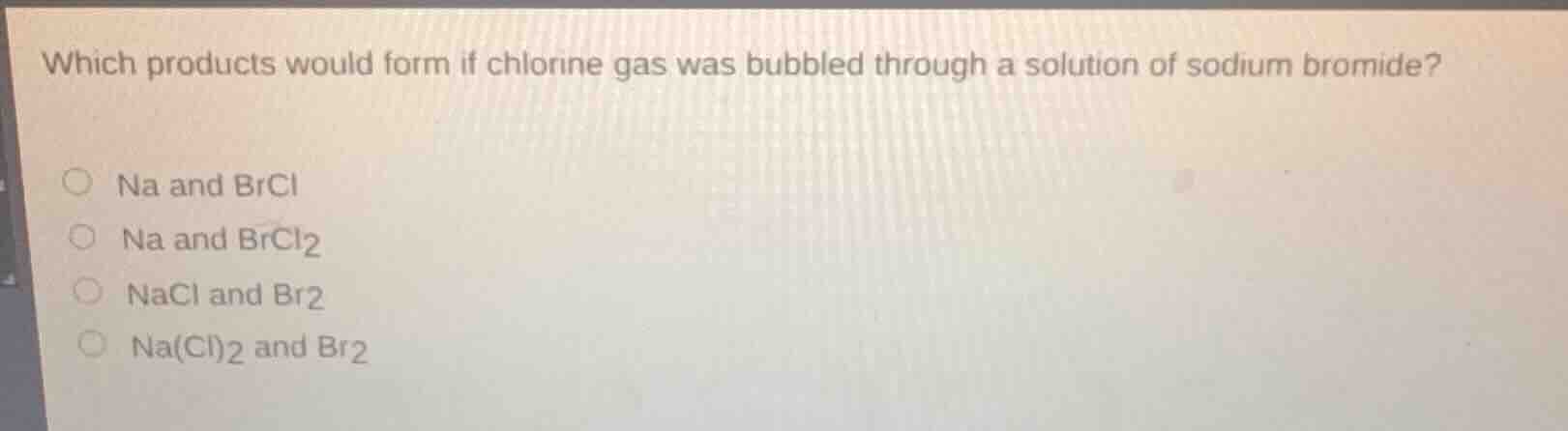
Question
which products would form if chlorine gas was bubbled through a solution of sodium bromide?
○ na and brcl
○ na and brcl₂
○ nacl and br₂
○ na(cl)₂ and br₂
This is a single - replacement reaction. In the activity series of halogens, chlorine ($\ce{Cl_2}$) is more reactive than bromine. So, when chlorine gas is bubbled through a sodium bromide ($\ce{NaBr}$) solution, chlorine will replace bromine in sodium bromide. The chemical reaction is $\ce{Cl_2 + 2NaBr = 2NaCl + Br_2}$. So the products are sodium chloride ($\ce{NaCl}$) and bromine ($\ce{Br_2}$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. NaCl and Br₂