QUESTION IMAGE
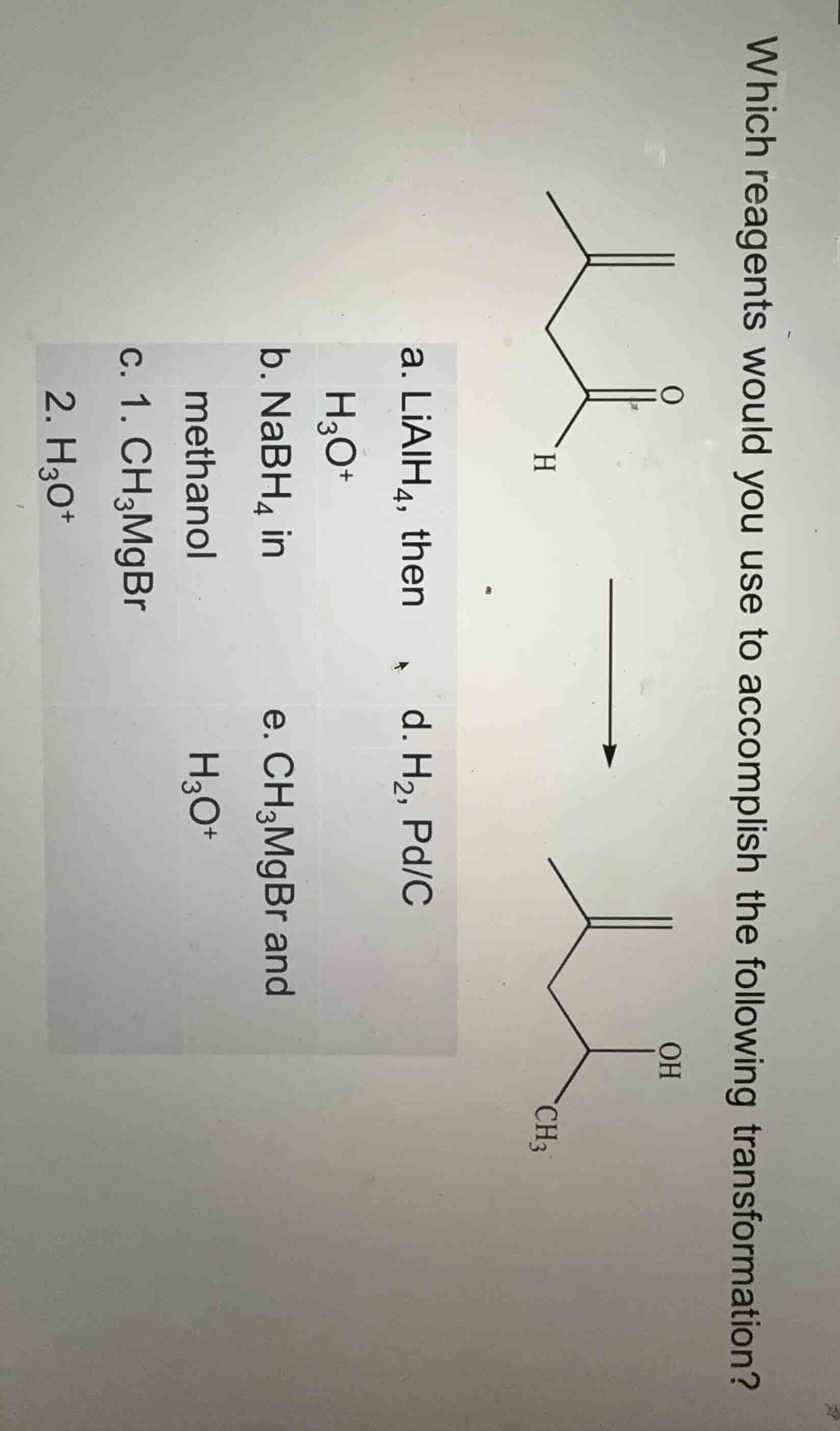
Question
which reagents would you use to accomplish the following transformation?
a. lialh₄, then h₃o⁺
b. nabh₄ in methanol
c. 1. ch₃mgbr 2. h₃o⁺
d. h₂, pd/c
e. ch₃mgbr and h₃o⁺
The starting material is an aldehyde (with a carbonyl group \( \text{C}=\text{O} \)) and we need to convert it to a secondary alcohol (with \( \text{-OH} \) and an added methyl group, \( \text{-CH}_3 \)). Let's analyze each option:
Step 1: Analyze Option a (\( \text{LiAlH}_4 \), then \( \text{H}_3\text{O}^+ \))
\( \text{LiAlH}_4 \) is a strong reducing agent for carbonyls. It reduces aldehydes to primary alcohols (adds \( \text{-H} \) to the carbonyl carbon), not secondary alcohols with a methyl group. So this is incorrect.
Step 2: Analyze Option b (\( \text{NaBH}_4 \) in methanol, then \( \text{H}_3\text{O}^+ \))
\( \text{NaBH}_4 \) is a mild reducing agent for carbonyls. It also reduces aldehydes to primary alcohols (adds \( \text{-H} \)), not introducing a methyl group. So this is incorrect.
Step 3: Analyze Option c (1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \))
Grignard reagents (\( \text{CH}_3\text{MgBr} \)) react with aldehydes. The \( \text{CH}_3^- \) (from \( \text{CH}_3\text{MgBr} \)) attacks the carbonyl carbon of the aldehyde. After protonation with \( \text{H}_3\text{O}^+ \), the carbonyl group (\( \text{C}=\text{O} \)) is converted to \( \text{-OH} \), and a methyl group is added to the carbonyl carbon, forming a secondary alcohol (since the original aldehyde carbonyl carbon now has a methyl, a hydrogen, and the -OH, plus the rest of the chain). This matches the desired product.
Step 4: Analyze Option d (\( \text{H}_2 \), \( \text{Pd/C} \))
\( \text{H}_2 \) with \( \text{Pd/C} \) is a hydrogenation catalyst, which reduces alkenes or alkynes, not carbonyl groups. The carbonyl group in the aldehyde would remain, so this is incorrect.
Step 5: Analyze Option e (1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \)) – Wait, Option c and e? Wait, looking back, Option c is 1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \), and Option e is same? Wait, maybe a typo, but assuming Option c is 1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \), and that's the Grignard reaction which adds a methyl group to the aldehyde carbonyl, forming a secondary alcohol. The other options don't add a methyl group. So the correct reagent is 1. \( \text{CH}_3\text{MgBr} \), then 2. \( \text{H}_3\text{O}^+ \), which is Option c (or e, but likely c as per the options). Wait, the options: c is 1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \); e is \( \text{CH}_3\text{MgBr} \) and \( \text{H}_3\text{O}^+ \) (maybe same as c). But the key is Grignard reagent (methyl Grignard) with an aldehyde gives a secondary alcohol with a methyl group.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c. 1. \( \text{CH}_3\text{MgBr} \), 2. \( \text{H}_3\text{O}^+ \)