QUESTION IMAGE
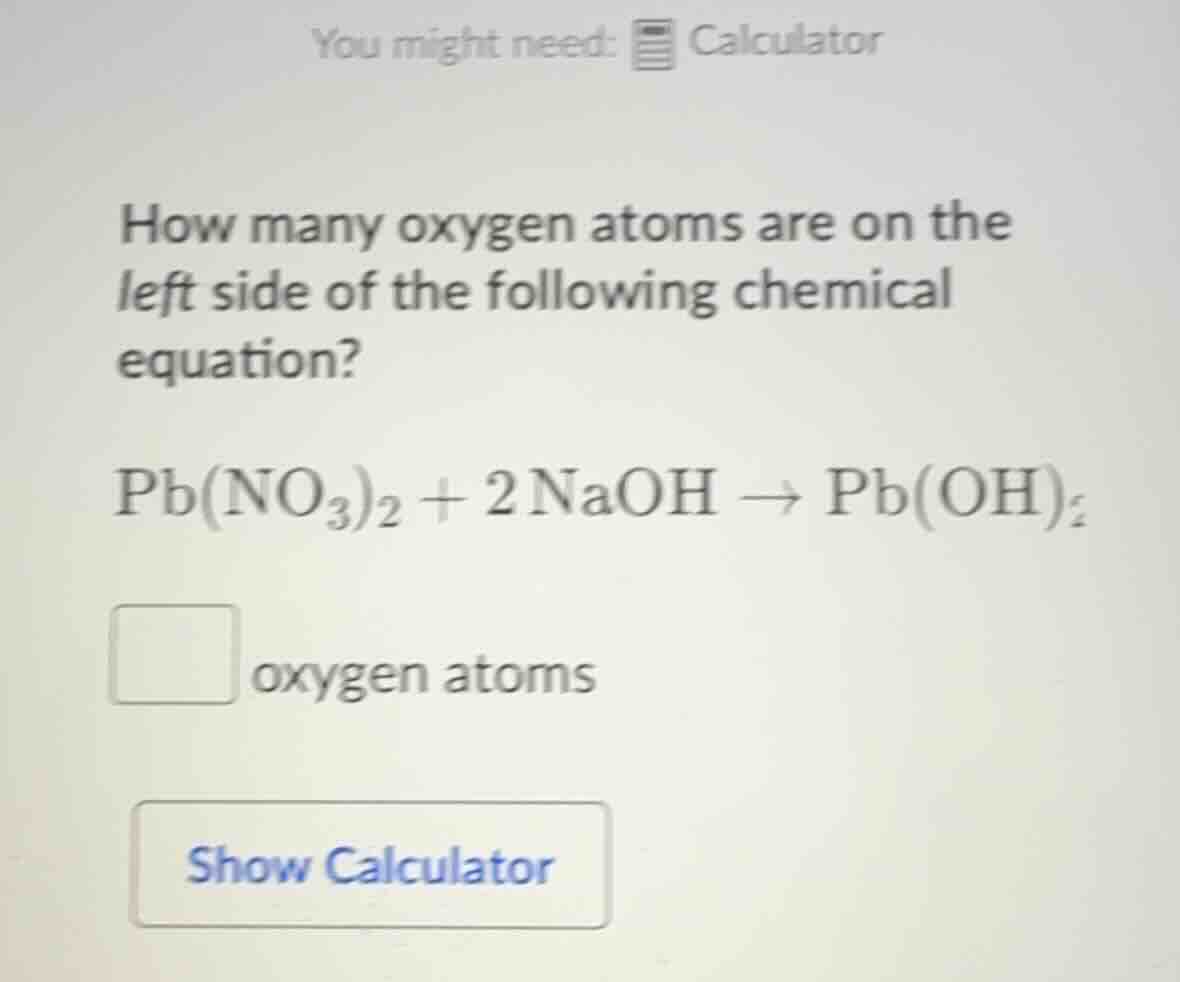
Question
you might need: calculator
how many oxygen atoms are on the left side of the following chemical equation?
pb(no₃)₂ + 2naoh → pb(oh)₂
□ oxygen atoms
show calculator
Step1: Analyze \( \text{Pb(NO}_3\text{)}_2 \)
In \( \text{Pb(NO}_3\text{)}_2 \), the subscript of \( \text{NO}_3 \) is 3, and it is multiplied by 2 (from the outer subscript). So oxygen atoms here: \( 3\times2 = 6 \).
Step2: Analyze \( 2\text{NaOH} \)
In \( \text{NaOH} \), there is 1 oxygen atom. The coefficient is 2, so oxygen atoms here: \( 1\times2 = 2 \).
Step3: Sum the oxygen atoms
Total oxygen atoms on the left: \( 6 + 2 = 8 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
8