QUESTION IMAGE
Question
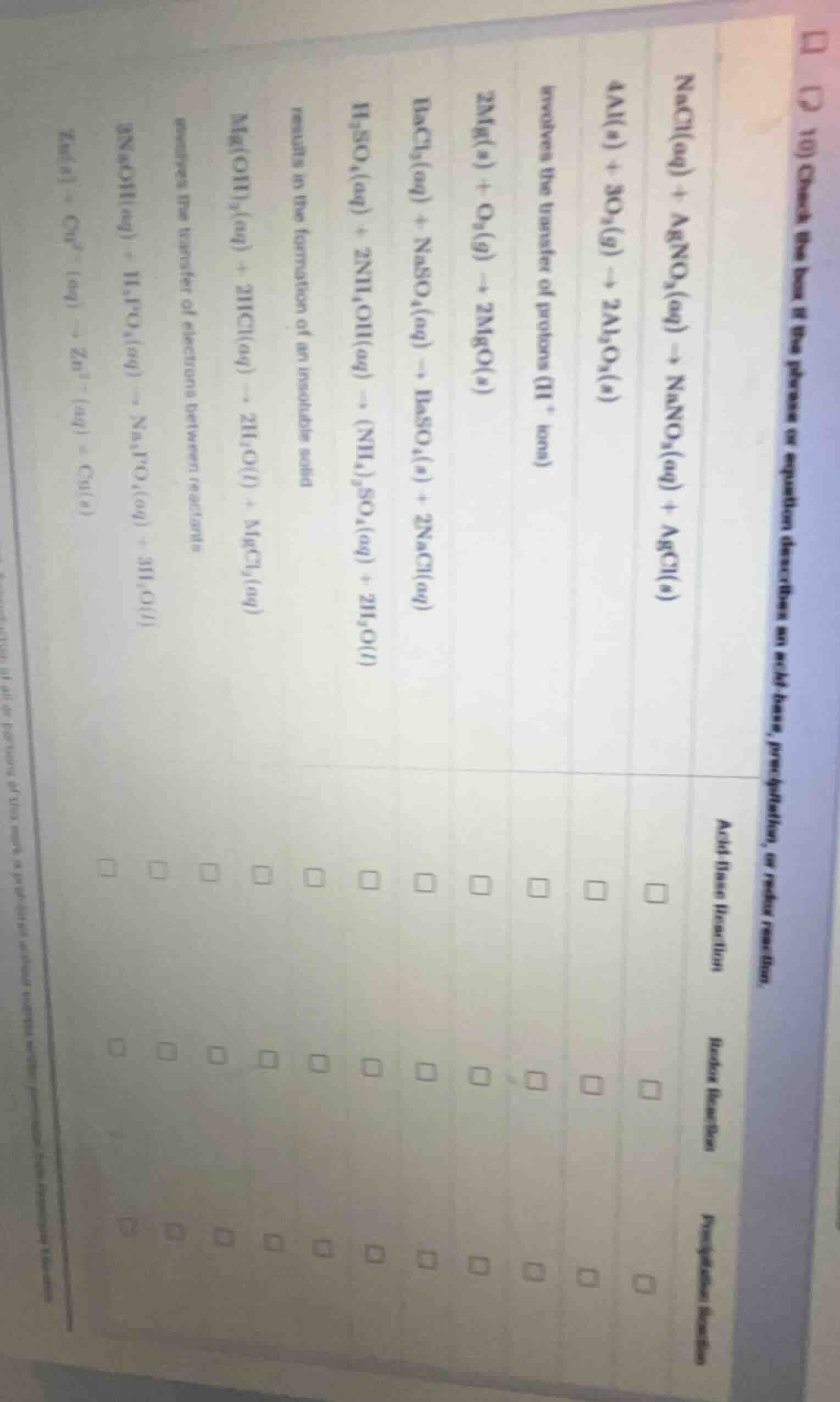
- check the box if the phrase or equation describes an acid-base, precipitation, or redox reaction.
acid-base reaction
precipitation reaction
redox reaction
nacl(aq) + agno₃(aq) → nano₃(aq) + agcl(s)
4al(s) + 3o₂(g) → 2al₂o₃(s)
involves the transfer of protons (h⁺ ions)
2mg(s) + o₂(g) → 2mgo(s)
bacl₂(aq) + na₂so₄(aq) → baso₄(s) + 2nacl(aq)
h₂so₄(aq) + 2nh₄oh(aq) → (nh₄)₂so₄(aq) + 2h₂o(l)
results in the formation of an insoluble solid
mg(oh)₂(aq) + 2hcl(aq) → 2h₂o(l) + mgcl₂(aq)
involves the transfer of electrons between reactants
3naoh(aq) + h₃po₄(aq) → na₃po₄(aq) + 3h₂o(l)
zn(s) + cu²⁺(aq) → zn²⁺(aq) + cu(s)
Step1: Classify first reaction
$\text{NaCl}(aq) + \text{AgNO}_3(aq)
ightarrow \text{NaNO}_3(aq) + \text{AgCl}(s)$: Forms insoluble solid $\text{AgCl}$, so it is a precipitation reaction.
Step2: Classify second reaction
$4\text{Al}(s) + 3\text{O}_2(g)
ightarrow 2\text{Al}_2\text{O}_3(s)$: Al (0) → Al³⁺ (+3), O (0) → O²⁻ (-2); electron transfer occurs, so it is a redox reaction.
Step3: Classify proton transfer phrase
"Involves transfer of protons ($\text{H}^+$ ions)": This is the definition of an acid-base reaction.
Step4: Classify fourth reaction
$2\text{Mg}(s) + \text{O}_2(g)
ightarrow 2\text{MgO}(s)$: Mg (0) → Mg²⁺ (+2), O (0) → O²⁻ (-2); electron transfer occurs, so it is a redox reaction.
Step5: Classify fifth reaction
$\text{BaCl}_2(aq) + \text{Na}_2\text{SO}_4(aq)
ightarrow \text{BaSO}_4(s) + 2\text{NaCl}(aq)$: Forms insoluble solid $\text{BaSO}_4$, so it is a precipitation reaction.
Step6: Classify sixth reaction
$\text{H}_2\text{SO}_4(aq) + 2\text{NH}_4\text{OH}(aq)
ightarrow (\text{NH}_4)_2\text{SO}_4(aq) + 2\text{H}_2\text{O}(l)$: $\text{H}^+$ from $\text{H}_2\text{SO}_4$ transfers to $\text{OH}^-$ from $\text{NH}_4\text{OH}$ to form $\text{H}_2\text{O}$, so it is an acid-base reaction.
Step7: Classify insoluble solid phrase
"Results in formation of an insoluble solid": This is the definition of a precipitation reaction.
Step8: Classify eighth reaction
$\text{Mg(OH)}_2(aq) + 2\text{HCl}(aq)
ightarrow 2\text{H}_2\text{O}(l) + \text{MgCl}_2(aq)$: $\text{H}^+$ from $\text{HCl}$ transfers to $\text{OH}^-$ from $\text{Mg(OH)}_2$ to form $\text{H}_2\text{O}$, so it is an acid-base reaction.
Step9: Classify electron transfer phrase
"Involves transfer of electrons between reactants": This is the definition of a redox reaction.
Step10: Classify tenth reaction
$3\text{NaOH}(aq) + \text{H}_3\text{PO}_4(aq)
ightarrow \text{Na}_3\text{PO}_4(aq) + 3\text{H}_2\text{O}(l)$: $\text{H}^+$ from $\text{H}_3\text{PO}_4$ transfers to $\text{OH}^-$ from $\text{NaOH}$ to form $\text{H}_2\text{O}$, so it is an acid-base reaction.
Step11: Classify eleventh reaction
$\text{Zn}(s) + \text{Cu}^{2+}(aq)
ightarrow \text{Zn}^{2+}(aq) + \text{Cu}(s)$: Zn (0) → Zn²⁺ (+2), Cu²⁺ (+2) → Cu (0); electron transfer occurs, so it is a redox reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{\text{NaCl}(aq) + \text{AgNO}_3(aq)
ightarrow \text{NaNO}_3(aq) + \text{AgCl}(s)}$: Check Precipitation Reaction box
- $\boldsymbol{4\text{Al}(s) + 3\text{O}_2(g)
ightarrow 2\text{Al}_2\text{O}_3(s)}$: Check Redox Reaction box
- $\boldsymbol{\text{Involves the transfer of protons (H}^+ \text{ ions)}}$: Check Acid-Base Reaction box
- $\boldsymbol{2\text{Mg}(s) + \text{O}_2(g)
ightarrow 2\text{MgO}(s)}$: Check Redox Reaction box
- $\boldsymbol{\text{BaCl}_2(aq) + \text{Na}_2\text{SO}_4(aq)
ightarrow \text{BaSO}_4(s) + 2\text{NaCl}(aq)}$: Check Precipitation Reaction box
- $\boldsymbol{\text{H}_2\text{SO}_4(aq) + 2\text{NH}_4\text{OH}(aq)
ightarrow (\text{NH}_4)_2\text{SO}_4(aq) + 2\text{H}_2\text{O}(l)}$: Check Acid-Base Reaction box
- $\boldsymbol{\text{Results in the formation of an insoluble solid}}$: Check Precipitation Reaction box
- $\boldsymbol{\text{Mg(OH)}_2(aq) + 2\text{HCl}(aq)
ightarrow 2\text{H}_2\text{O}(l) + \text{MgCl}_2(aq)}$: Check Acid-Base Reaction box
- $\boldsymbol{\text{Involves the transfer of electrons between reactants}}$: Check Redox Reaction box
- $\boldsymbol{3\text{NaOH}(aq) + \text{H}_3\text{PO}_4(aq)
ightarrow \text{Na}_3\text{PO}_4(aq) + 3\text{H}_2\text{O}(l)}$: Check Acid-Base Reaction box
- $\boldsymbol{\text{Zn}(s) + \text{Cu}^{2+}(aq)
ightarrow \text{Zn}^{2+}(aq) + \text{Cu}(s)}$: Check Redox Reaction box