QUESTION IMAGE
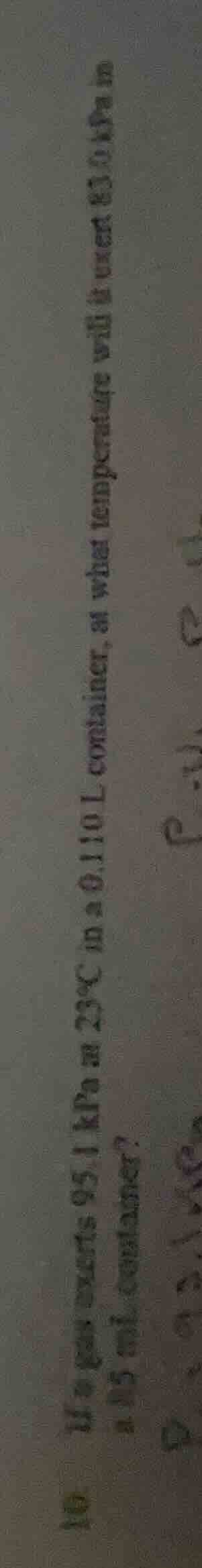
Question
- a gas exerts 95.1 kpa at 25°c in a 0.110 l container. at what temperature will it exert 63.0 kpa in a 0.35 ml container?
Step1: List given values
$P_1=95.1\ \text{kPa}$, $V_1=0.110\ \text{L}$, $T_1=23^\circ\text{C}=23+273.15=296.15\ \text{K}$
$P_2=83.0\ \text{kPa}$, $V_2=85\ \text{mL}=0.085\ \text{L}$, $T_2=?$
Step2: Use combined gas law
The combined gas law is $\frac{P_1V_1}{T_1}=\frac{P_2V_2}{T_2}$. Rearrange to solve for $T_2$:
$T_2=\frac{P_2V_2T_1}{P_1V_1}$
Step3: Substitute values into formula
$T_2=\frac{83.0\ \text{kPa} \times 0.085\ \text{L} \times 296.15\ \text{K}}{95.1\ \text{kPa} \times 0.110\ \text{L}}$
Step4: Calculate the result
First compute numerator: $83.0 \times 0.085 \times 296.15 = 83.0 \times 25.17275 = 2089.33825$
Then compute denominator: $95.1 \times 0.110 = 10.461$
$T_2=\frac{2089.33825}{10.461} \approx 199.7\ \text{K}$
Convert to Celsius: $T_2=199.7-273.15 \approx -73.5^\circ\text{C}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The temperature is approximately $\boldsymbol{200\ \text{K}}$ (or $\boldsymbol{-73^\circ\text{C}}$)