QUESTION IMAGE
Question
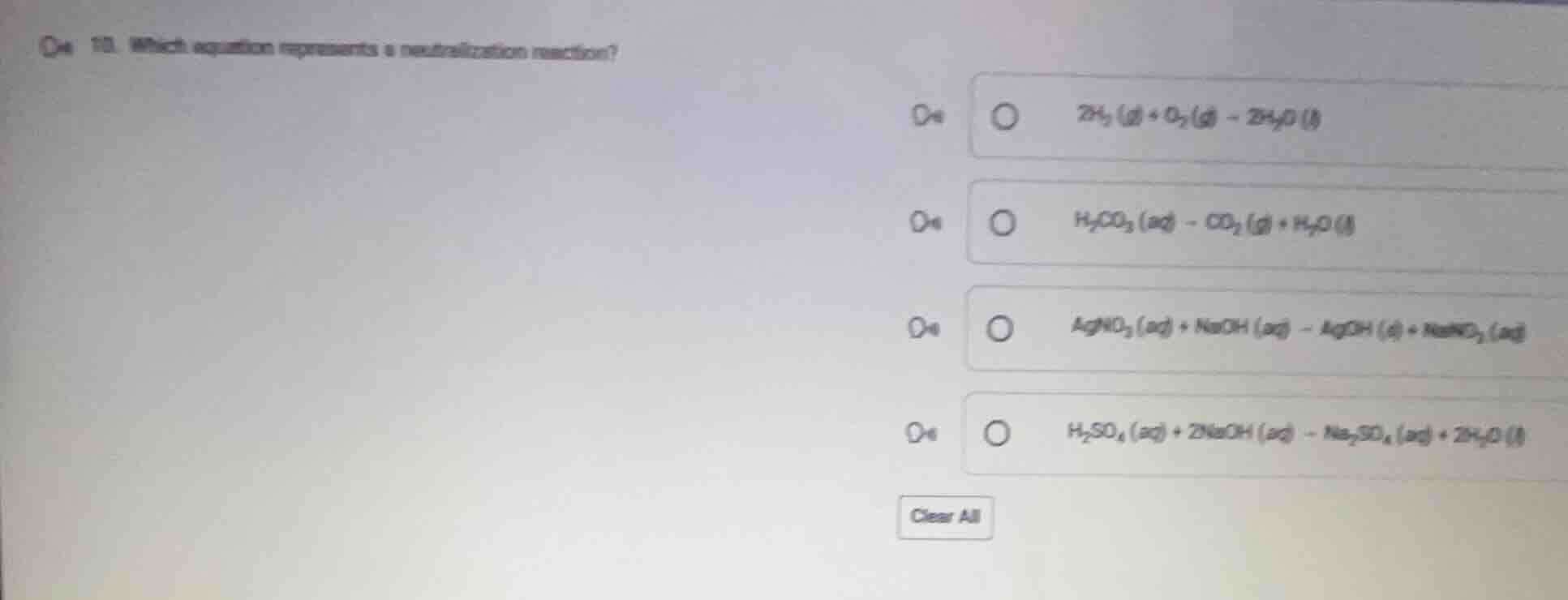
- which equation represents a neutralization reaction?
$2h_2(g) + o_2(g) \
ightarrow 2h_2o(l)$
$h_2co_3(aq) \
ightarrow co_2(g) + h_2o(l)$
$agno_3(aq) + naoh(aq) \
ightarrow agoh(s) + nano_3(aq)$
$h_2so_4(aq) + 2naoh(aq) \
ightarrow na_2so_4(aq) + 2h_2o(l)$
Step1: Define neutralization reaction
A neutralization reaction is a reaction between an acid and a base that produces a salt and water.
Step2: Analyze Option A
$\ce{2H2 (g) + O2 (g) -> 2H2O (l)}$ is a synthesis reaction of water, not acid-base.
Step3: Analyze Option B
$\ce{H2CO3 (aq) -> CO2 (g) + H2O (l)}$ is a decomposition of a carbonic acid, not acid-base.
Step4: Analyze Option C
$\ce{AgNO3 (aq) + NaOH (aq) -> AgOH (s) + NaNO3 (aq)}$ is a double displacement reaction forming a precipitate, not acid-base.
Step5: Analyze Option D
$\ce{H2SO4 (aq)}$ is an acid, $\ce{2NaOH (aq)}$ is a base. They react to form $\ce{Na2SO4 (aq)}$ (a salt) and $\ce{2H2O (l)}$ (water), which fits the neutralization definition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\ce{H2SO4 (aq) + 2NaOH (aq) -> Na2SO4 (aq) + 2H2O (l)}$