QUESTION IMAGE
Question
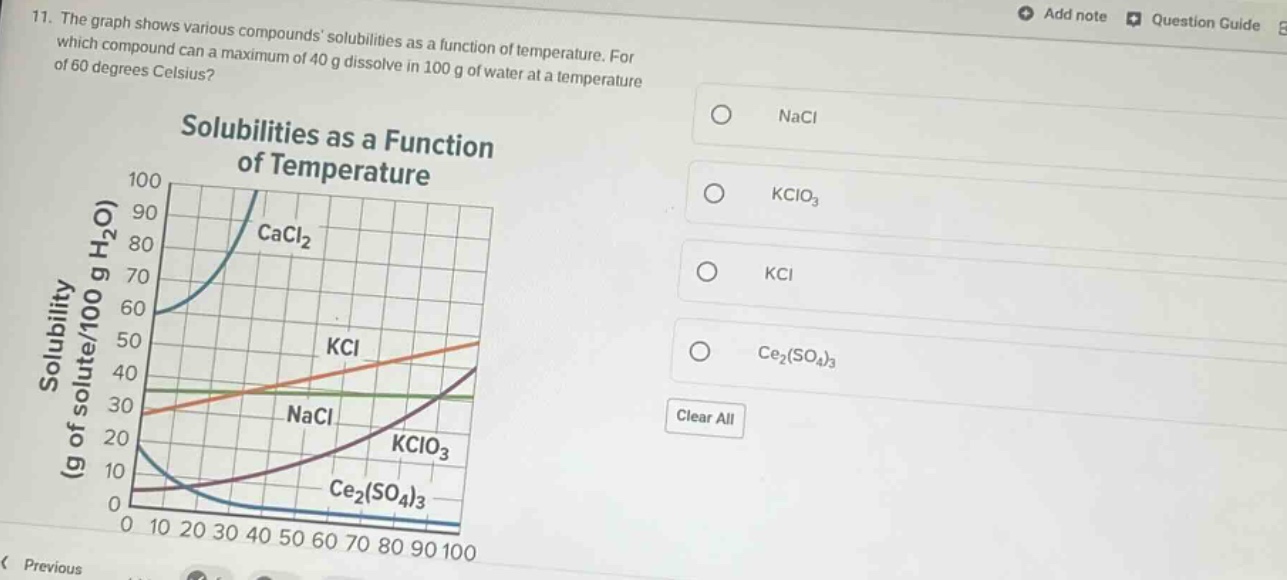
- the graph shows various compounds solubilities as a function of temperature. for which compound can a maximum of 40 g dissolve in 100 g of water at a temperature of 60 degrees celsius? solubilities as a function of temperature \
$$\begin{tabular}{|c|c|c|c|c|c|c|c|c|c|c|} \\hline & 0 & 10 & 20 & 30 & 40 & 50 & 60 & 70 & 80 & 90 & 100 \\\\ \\hline solubility (g of solute/100 g h₂o) & & & & & & & & & & & \\\\ \\hline cacl₂ & 60 & & & & & & & & & & \\\\ \\hline kcl & 30 & & & & & & & & & & 50 \\\\ \\hline nacl & 30 & & & & & & & & & & \\\\ \\hline kclo₃ & 10 & & & & & & & & & & 40 \\\\ \\hline ce₂(so₄)₃ & 20 & & & & & & & & & & 0 \\\\ \\hline \\end{tabular}$$
options: \bigcirc nacl \bigcirc kclo₃ \bigcirc kcl \bigcirc ce₂(so₄)₃ clear all
Step1: Analyze the solubility graph at 60°C
We need to check the solubility (g of solute/100 g H₂O) of each compound at 60°C from the graph.
Step2: Check each compound's solubility
- For NaCl: The solubility curve of NaCl at 60°C is below 40 g/100 g H₂O.
- For KClO₃: The solubility curve of KClO₃ at 60°C is below 40 g/100 g H₂O.
- For KCl: The solubility curve of KCl at 60°C is around or above 40 g/100 g H₂O (from the graph, the KCl curve at 60°C is above the 40 g line).
- For Ce₂(SO₄)₃: The solubility curve of Ce₂(SO₄)₃ at 60°C is below 40 g/100 g H₂O (and decreasing).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. KCl