QUESTION IMAGE
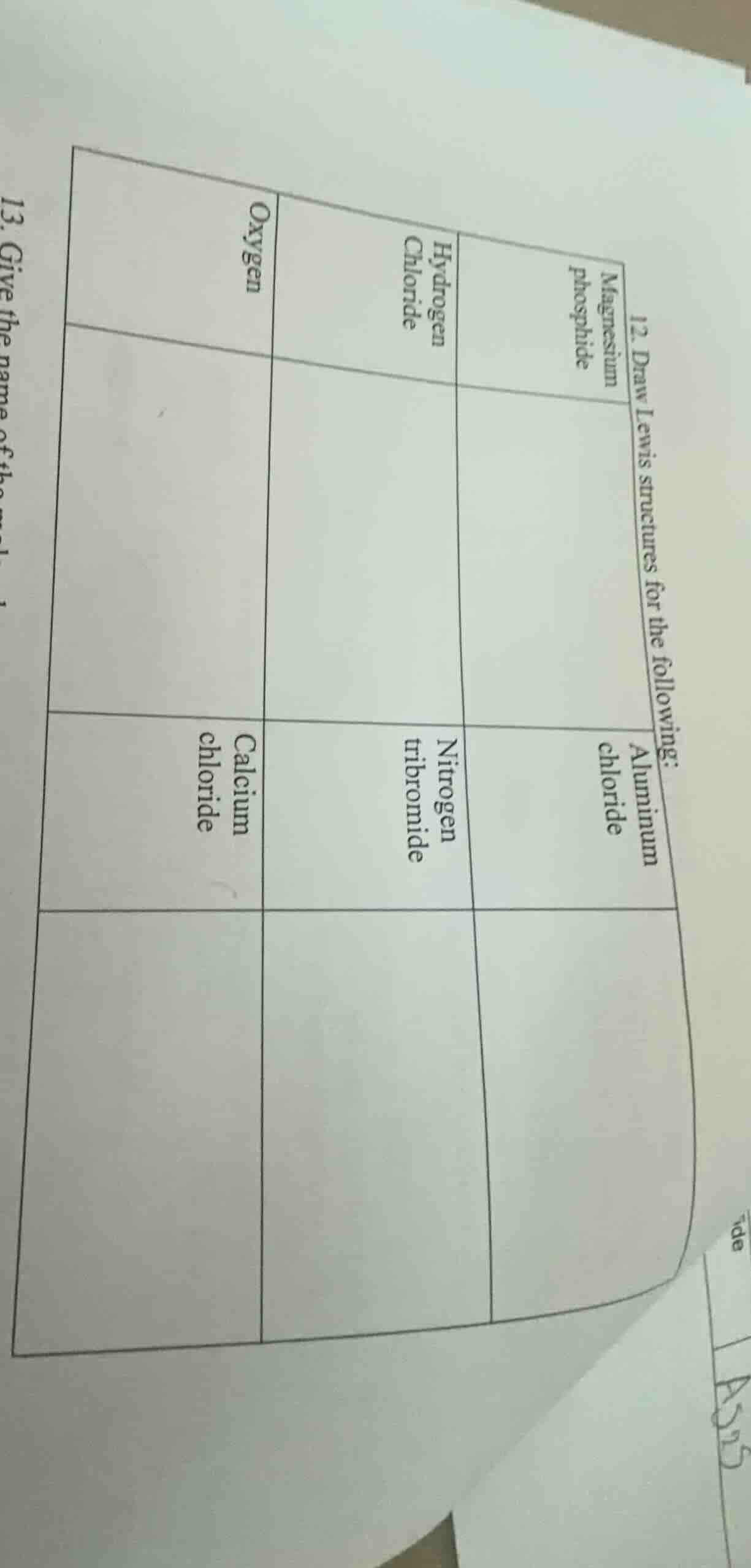
Question
- draw lewis structures for the following:
magnesium phosphide
aluminum chloride
hydrogen chloride
oxygen
nitrogen tribromide
calcium chloride
Response
To solve this, we determine the chemical formulas by balancing the charges of the ions.
Magnesium phosphide
- Magnesium ion: \( \text{Mg}^{2+} \)
- Phosphide ion: \( \text{P}^{3-} \)
- To balance charges, find the least common multiple of 2 and 3, which is 6. So we need 3 \( \text{Mg}^{2+} \) and 2 \( \text{P}^{3-} \).
- Formula: \( \text{Mg}_3\text{P}_2 \)
Aluminum chloride
- Aluminum ion: \( \text{Al}^{3+} \)
- Chloride ion: \( \text{Cl}^- \)
- To balance, we need 1 \( \text{Al}^{3+} \) and 3 \( \text{Cl}^- \).
- Formula: \( \text{AlCl}_3 \)
Hydrogen chloride
- Hydrogen ion: \( \text{H}^+ \)
- Chloride ion: \( \text{Cl}^- \)
- Charges balance 1:1.
- Formula: \( \text{HCl} \)
Nitrogen tribromide
- Nitrogen: \( \text{N} \) (neutral, since it's a molecular compound with bromine)
- Bromide: \( \text{Br} \)
- Prefix "tri-" means 3 bromine atoms.
- Formula: \( \text{NBr}_3 \)
Oxygen (assuming it's a diatomic molecule, common form)
- Oxygen exists as \( \text{O}_2 \) in its elemental diatomic form.
Calcium chloride
- Calcium ion: \( \text{Ca}^{2+} \)
- Chloride ion: \( \text{Cl}^- \)
- To balance, 1 \( \text{Ca}^{2+} \) and 2 \( \text{Cl}^- \).
- Formula: \( \text{CaCl}_2 \)
Lewis Structures (brief for each):
- \( \text{Mg}_3\text{P}_2 \): Ionic compound, so we represent the ions. \( \text{Mg}^{2+} \) and \( \text{P}^{3-} \) with electron transfer (but Lewis for ionic is about ion arrangement, showing cations and anions).
- \( \text{AlCl}_3 \): Aluminum has 3 valence electrons, each Cl has 7. Al shares 3 electrons with 3 Cl atoms, forming 3 single bonds. Al: \( \cdot \text{Al} \cdot \) (3 valence), each Cl: \( :\ddot{\text{Cl}}: \), so Al is bonded to 3 Cl, each Cl has 3 lone pairs.
- \( \text{HCl} \): Single bond between H (1 valence) and Cl (7 valence). H: \( \text{H} \), Cl: \( :\ddot{\text{Cl}}: \), bonded as \( \text{H}-\text{Cl} \) with Cl having 3 lone pairs.
- \( \text{NBr}_3 \): Nitrogen has 5 valence electrons, each Br has 7. N forms 3 single bonds with Br, using 3 electrons, leaving 1 lone pair on N. Each Br has 3 lone pairs. Structure: \( :\text{N}-\text{Br}: \), \( :\text{N}-\text{Br}: \), \( :\text{N}-\text{Br}: \) (with N having a lone pair above).
- \( \text{O}_2 \): Double bond between two O atoms, each with 2 lone pairs. \( :\ddot{\text{O}}=\ddot{\text{O}}: \)
- \( \text{CaCl}_2 \): Ionic, \( \text{Ca}^{2+} \) and two \( \text{Cl}^- \) ions, showing electron transfer (Ca loses 2, each Cl gains 1).
Final Formulas and Lewis Summary:
| Compound | Formula | Lewis Structure (Brief) |
|---|---|---|
| Aluminum chloride | \( \text{AlCl}_3 \) | \( \text{Al} \) bonded to 3 \( \text{Cl} \), each \( \text{Cl} \) has 3 lone pairs. |
| Hydrogen chloride | \( \text{HCl} \) | Single bond \( \text{H}-\text{Cl} \), \( \text{Cl} \) has 3 lone pairs. |
| Nitrogen tribromide | \( \text{NBr}_3 \) | \( \text{N} \) bonded to 3 \( \text{Br} \), \( \text{N} \) has 1 lone pair, each \( \text{Br} \) has 3. |
| Oxygen | \( \text{O}_2 \) | Double bond \( :\ddot{\text{O}}=\ddot{\text{O}}: \), each O has 2 lone pairs. |
| Calcium chloride | \( \text{CaCl}_2 \) | Ionic: \( \text… |
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we determine the chemical formulas by balancing the charges of the ions.
Magnesium phosphide
- Magnesium ion: \( \text{Mg}^{2+} \)
- Phosphide ion: \( \text{P}^{3-} \)
- To balance charges, find the least common multiple of 2 and 3, which is 6. So we need 3 \( \text{Mg}^{2+} \) and 2 \( \text{P}^{3-} \).
- Formula: \( \text{Mg}_3\text{P}_2 \)
Aluminum chloride
- Aluminum ion: \( \text{Al}^{3+} \)
- Chloride ion: \( \text{Cl}^- \)
- To balance, we need 1 \( \text{Al}^{3+} \) and 3 \( \text{Cl}^- \).
- Formula: \( \text{AlCl}_3 \)
Hydrogen chloride
- Hydrogen ion: \( \text{H}^+ \)
- Chloride ion: \( \text{Cl}^- \)
- Charges balance 1:1.
- Formula: \( \text{HCl} \)
Nitrogen tribromide
- Nitrogen: \( \text{N} \) (neutral, since it's a molecular compound with bromine)
- Bromide: \( \text{Br} \)
- Prefix "tri-" means 3 bromine atoms.
- Formula: \( \text{NBr}_3 \)
Oxygen (assuming it's a diatomic molecule, common form)
- Oxygen exists as \( \text{O}_2 \) in its elemental diatomic form.
Calcium chloride
- Calcium ion: \( \text{Ca}^{2+} \)
- Chloride ion: \( \text{Cl}^- \)
- To balance, 1 \( \text{Ca}^{2+} \) and 2 \( \text{Cl}^- \).
- Formula: \( \text{CaCl}_2 \)
Lewis Structures (brief for each):
- \( \text{Mg}_3\text{P}_2 \): Ionic compound, so we represent the ions. \( \text{Mg}^{2+} \) and \( \text{P}^{3-} \) with electron transfer (but Lewis for ionic is about ion arrangement, showing cations and anions).
- \( \text{AlCl}_3 \): Aluminum has 3 valence electrons, each Cl has 7. Al shares 3 electrons with 3 Cl atoms, forming 3 single bonds. Al: \( \cdot \text{Al} \cdot \) (3 valence), each Cl: \( :\ddot{\text{Cl}}: \), so Al is bonded to 3 Cl, each Cl has 3 lone pairs.
- \( \text{HCl} \): Single bond between H (1 valence) and Cl (7 valence). H: \( \text{H} \), Cl: \( :\ddot{\text{Cl}}: \), bonded as \( \text{H}-\text{Cl} \) with Cl having 3 lone pairs.
- \( \text{NBr}_3 \): Nitrogen has 5 valence electrons, each Br has 7. N forms 3 single bonds with Br, using 3 electrons, leaving 1 lone pair on N. Each Br has 3 lone pairs. Structure: \( :\text{N}-\text{Br}: \), \( :\text{N}-\text{Br}: \), \( :\text{N}-\text{Br}: \) (with N having a lone pair above).
- \( \text{O}_2 \): Double bond between two O atoms, each with 2 lone pairs. \( :\ddot{\text{O}}=\ddot{\text{O}}: \)
- \( \text{CaCl}_2 \): Ionic, \( \text{Ca}^{2+} \) and two \( \text{Cl}^- \) ions, showing electron transfer (Ca loses 2, each Cl gains 1).
Final Formulas and Lewis Summary:
| Compound | Formula | Lewis Structure (Brief) |
|---|---|---|
| Aluminum chloride | \( \text{AlCl}_3 \) | \( \text{Al} \) bonded to 3 \( \text{Cl} \), each \( \text{Cl} \) has 3 lone pairs. |
| Hydrogen chloride | \( \text{HCl} \) | Single bond \( \text{H}-\text{Cl} \), \( \text{Cl} \) has 3 lone pairs. |
| Nitrogen tribromide | \( \text{NBr}_3 \) | \( \text{N} \) bonded to 3 \( \text{Br} \), \( \text{N} \) has 1 lone pair, each \( \text{Br} \) has 3. |
| Oxygen | \( \text{O}_2 \) | Double bond \( :\ddot{\text{O}}=\ddot{\text{O}}: \), each O has 2 lone pairs. |
| Calcium chloride | \( \text{CaCl}_2 \) | Ionic: \( \text{Ca}^{2+} \) and \( 2\text{Cl}^- \) in lattice. |
(Note: For a more detailed Lewis structure drawing, we use dots for valence electrons, but the above gives the key bonding and lone pair details.)