QUESTION IMAGE
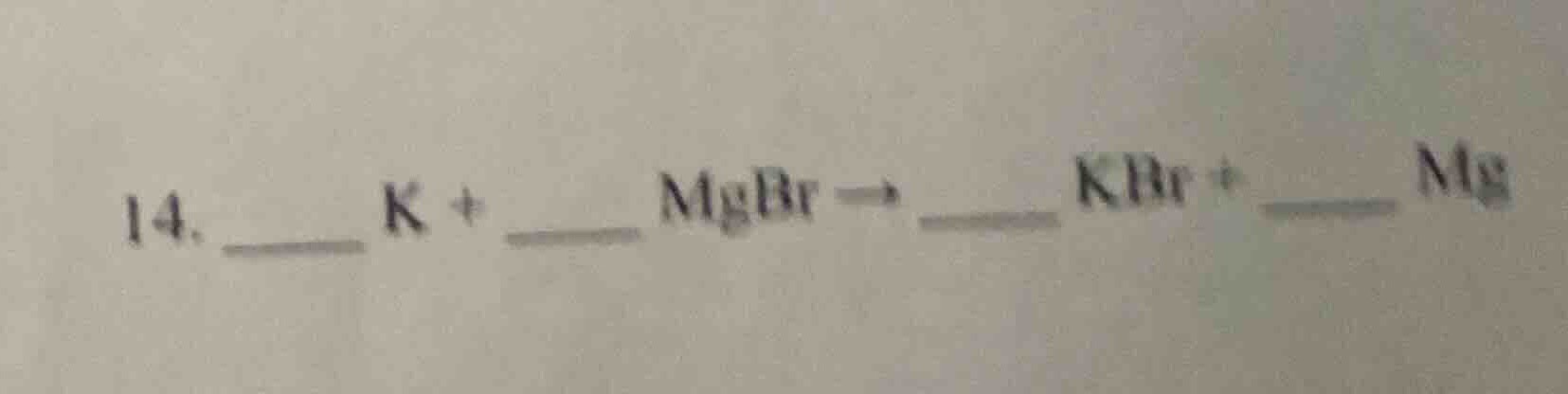
Question
- __ k + mgbr → kbr + __ mg
Step1: Balance Br atoms
On the left side, we have 1 Br from \( \text{MgBr} \), and on the right side, we have 1 Br from \( \text{KBr} \). So Br is balanced for now.
Step2: Balance K atoms
Let's look at K. On the left, we have \( x \) K atoms, and on the right, we have \( z \) K atoms from \( \text{KBr} \). Let's assume the coefficient of \( \text{K} \) is \( x \), \( \text{MgBr} \) is \( y \), \( \text{KBr} \) is \( z \), and \( \text{Mg} \) is \( w \).
From Br: \( y = z \) (since each \( \text{MgBr} \) has 1 Br and each \( \text{KBr} \) has 1 Br).
From K: \( x = z \) (since each \( \text{K} \) gives 1 K to \( \text{KBr} \)).
From Mg: \( y = w \) (since each \( \text{MgBr} \) has 1 Mg and each \( \text{Mg} \) is a product).
Let's start with \( y = 1 \). Then \( z = 1 \), \( x = 1 \), \( w = 1 \).
Check the equation: \( 1 \text{K} + 1 \text{MgBr}
ightarrow 1 \text{KBr} + 1 \text{Mg} \)
Now check all atoms:
- K: 1 on left, 1 on right.
- Mg: 1 on left, 1 on right.
- Br: 1 on left, 1 on right.
So the balanced equation is \( \boldsymbol{1} \text{K} + \boldsymbol{1} \text{MgBr}
ightarrow \boldsymbol{1} \text{KBr} + \boldsymbol{1} \text{Mg} \) (Note: Wait, actually, the formula for magnesium bromide should be \( \text{MgBr}_2 \) (since Mg has a +2 charge and Br has a -1 charge, so correct formula is \( \text{MgBr}_2 \)). Maybe there was a typo, but assuming the given formula is \( \text{MgBr} \) (even though it's incorrect in reality), the balancing as per given formula is as above. If it's \( \text{MgBr}_2 \), then:
Correcting the formula to \( \text{MgBr}_2 \):
Step1: Balance Br atoms
Left: 2 Br (from \( \text{MgBr}_2 \)), Right: 1 Br (from \( \text{KBr} \)). So we need 2 \( \text{KBr} \) to balance Br. So \( z = 2 \).
Step2: Balance K atoms
Since \( z = 2 \), \( x = 2 \) (K on left).
Step3: Balance Mg atoms
Left: 1 Mg (from \( \text{MgBr}_2 \)), Right: \( w \) Mg. So \( w = 1 \).
Left: \( y = 1 \) ( \( \text{MgBr}_2 \) ), so Br: \( 2y = 2(1) = 2 \), Right: \( z = 2 \) ( \( \text{KBr} \) has 1 Br each, 2 KBr have 2 Br).
Now the equation: \( 2 \text{K} + 1 \text{MgBr}_2
ightarrow 2 \text{KBr} + 1 \text{Mg} \)
Check atoms:
- K: 2 on left, 2 on right.
- Mg: 1 on left, 1 on right.
- Br: 2 on left, 2 on right.
But the original problem has \( \text{MgBr} \), so assuming the given formula is correct (even though it's a wrong formula in chemistry, maybe a typo), the first balancing (with \( \text{MgBr} \)) gives coefficients 1,1,1,1.
But likely, it's a typo and should be \( \text{MgBr}_2 \). Let's confirm the common reaction: Potassium reacts with magnesium bromide. The correct formula for magnesium bromide is \( \text{MgBr}_2 \). So the correct balancing is:
\( 2 \text{K} + 1 \text{MgBr}_2
ightarrow 2 \text{KBr} + 1 \text{Mg} \)
But based on the given formula \( \text{MgBr} \) (incorrect), the coefficients are 1,1,1,1.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The coefficients are \( \boldsymbol{1} \) (for K), \( \boldsymbol{1} \) (for MgBr), \( \boldsymbol{1} \) (for KBr), and \( \boldsymbol{1} \) (for Mg) (assuming \( \text{MgBr} \) is the given formula, though it's chemically incorrect; if it's \( \text{MgBr}_2 \), the coefficients are 2, 1, 2, 1).