QUESTION IMAGE
Question
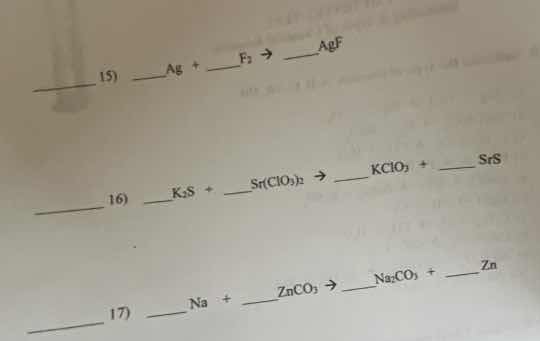
- _ag + _f₂ → ___agf
- _k₂s + _sr(clo₃)₂ → _kclo₃ + _srs
- _na + _znco₃ → _na₂co₃ + _zn
Step1: Balance equation 15: Ag + F₂ → AgF
First balance F: There are 2 F on left, so add coefficient 2 to AgF.
$\text{Ag} + \text{F}_2
ightarrow 2\text{AgF}$
Now balance Ag: There are 2 Ag on right, so add coefficient 2 to Ag.
$2\text{Ag} + \text{F}_2
ightarrow 2\text{AgF}$
Step2: Balance equation 16: K₂S + Sr(ClO₃)₂ → KClO₃ + SrS
Balance K: 2 K on left, add coefficient 2 to KClO₃.
$\text{K}_2\text{S} + \text{Sr(ClO}_3\text{)}_2
ightarrow 2\text{KClO}_3 + \text{SrS}$
All other atoms (S, Sr, Cl, O) are now balanced.
Step3: Balance equation 17: Na + ZnCO₃ → Na₂CO₃ + Zn
Balance Na: 2 Na on right, add coefficient 2 to Na.
$2\text{Na} + \text{ZnCO}_3
ightarrow \text{Na}_2\text{CO}_3 + \text{Zn}$
All other atoms (Zn, C, O) are now balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{Ag} + \text{F}_2
ightarrow 2\text{AgF}$
- $\text{K}_2\text{S} + \text{Sr(ClO}_3\text{)}_2
ightarrow 2\text{KClO}_3 + \text{SrS}$
- $2\text{Na} + \text{ZnCO}_3
ightarrow \text{Na}_2\text{CO}_3 + \text{Zn}$