QUESTION IMAGE
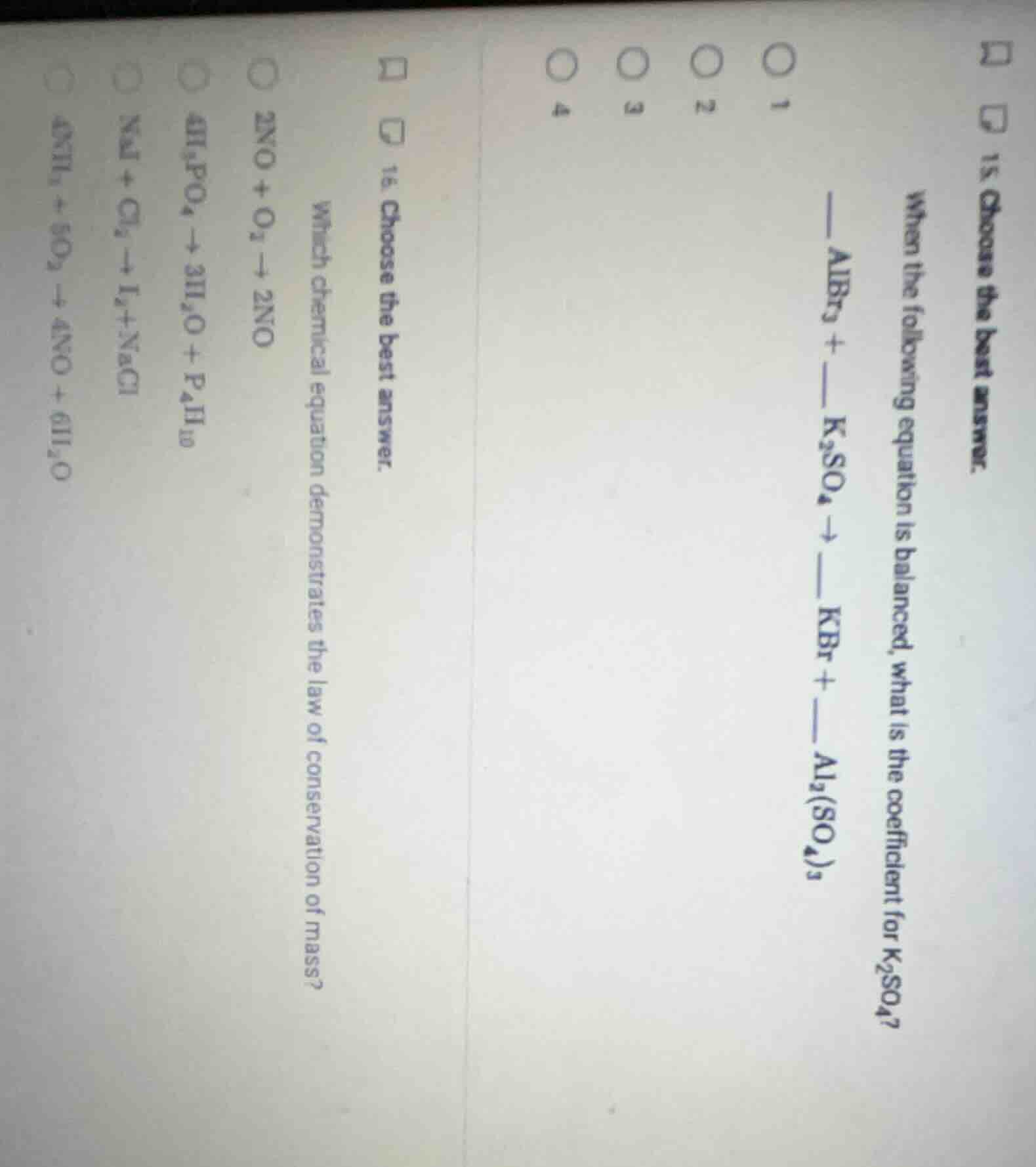
Question
- choose the best answer. when the following equation is balanced, what is the coefficient for k₂so₄? albr₃ + k₂so₄ → kbr + al₂(so₄)₃ 16. choose the best answer. which chemical equation demonstrates the law of conservation of mass? 2no + o₃ → 2no 4h₃po₄ → 3h₂o + p₄h₁₀ nai + cl₂ → i₂ + nacl 4nh₃ + 5o₂ → 4no + 6h₂o
Step1: Balance Al atoms
We have 2 Al on the right ($\text{Al}_2(\text{SO}_4)_3$), so add coefficient 2 to $\text{AlBr}_3$:
$2\text{AlBr}_3 + \_\_\text{K}_2\text{SO}_4
ightarrow \_\_\text{KBr} + \text{Al}_2(\text{SO}_4)_3$
Step2: Balance Br atoms
Now we have 6 Br on the left, so add coefficient 6 to $\text{KBr}$:
$2\text{AlBr}_3 + \_\_\text{K}_2\text{SO}_4
ightarrow 6\text{KBr} + \text{Al}_2(\text{SO}_4)_3$
Step3: Balance K atoms
We have 6 K on the right, so add coefficient 3 to $\text{K}_2\text{SO}_4$ (since $3\times2=6$ K):
$2\text{AlBr}_3 + 3\text{K}_2\text{SO}_4
ightarrow 6\text{KBr} + \text{Al}_2(\text{SO}_4)_3$
Step4: Verify $\text{SO}_4^{2-}$ ions
3 $\text{SO}_4^{2-}$ on left, 3 on right; all atoms balanced.
---
Step1: Check law of conservation of mass
Law requires equal atoms on both sides of a balanced equation.
Step2: Analyze Option 1
$2\text{NO} + \text{O}_2
ightarrow 2\text{NO}$: Left has 2 N, 4 O; right has 2 N, 2 O. Unbalanced.
Step3: Analyze Option 2
$4\text{H}_3\text{PO}_4
ightarrow 3\text{H}_2\text{O} + \text{P}_4\text{H}_{10}$: Left has 12 H, 4 P, 16 O; right has 16 H, 4 P, 3 O. Unbalanced.
Step4: Analyze Option 3
$\text{NaI} + \text{Cl}_2
ightarrow \text{I}_2+\text{NaCl}$: Left has 1 Na, 1 I, 2 Cl; right has 2 I, 1 Na, 1 Cl. Unbalanced.
Step5: Analyze Option 4
$4\text{NH}_3 + 5\text{O}_2
ightarrow 4\text{NO} + 6\text{H}_2\text{O}$: Left: 4 N, 12 H, 10 O; Right: 4 N, 12 H, 10 O. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- B. 3
- D. $4\text{NH}_3 + 5\text{O}_2
ightarrow 4\text{NO} + 6\text{H}_2\text{O}$