QUESTION IMAGE
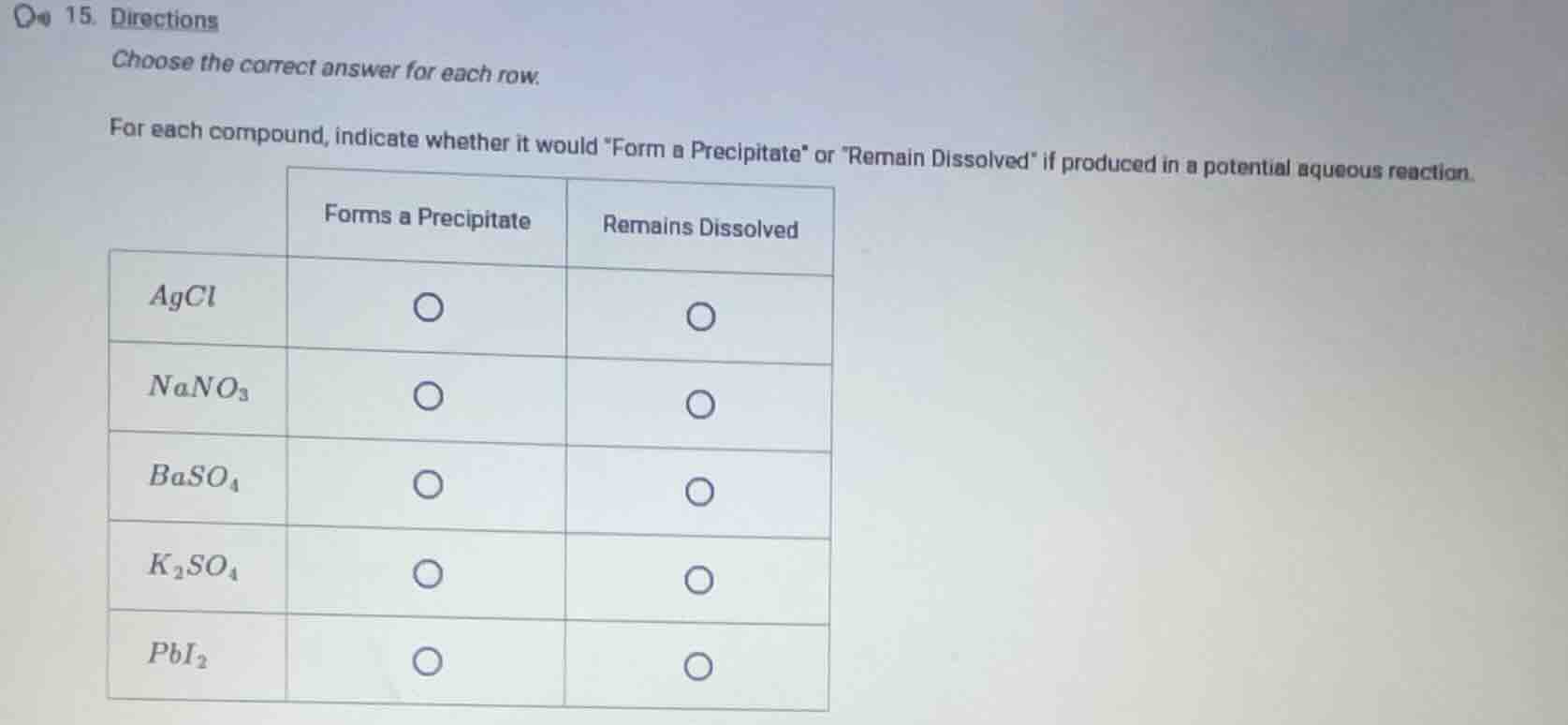
Question
- directions
choose the correct answer for each row.
for each compound, indicate whether it would \form a precipitate\ or
emain dissolved\ if produced in a potential aqueous reaction.
| forms a precipitate | remains dissolved | |
|---|---|---|
| $nano_3$ | $\bigcirc$ | $\bigcirc$ |
| $baso_4$ | $\bigcirc$ | $\bigcirc$ |
| $k_2so_4$ | $\bigcirc$ | $\bigcirc$ |
| $pbi_2$ | $\bigcirc$ | $\bigcirc$ |
Brief Explanations
- Silver chloride ($AgCl$) is an insoluble halide salt in water, so it forms a precipitate.
- All nitrate salts (including $NaNO_3$) are soluble in water, so it remains dissolved.
- Barium sulfate ($BaSO_4$) is one of the few insoluble sulfate salts, so it forms a precipitate.
- Salts containing group 1 metals (like $K^+$ in $K_2SO_4$) are soluble in water, so it remains dissolved.
- Lead(II) iodide ($PbI_2$) is an insoluble halide salt in water, so it forms a precipitate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $AgCl$: Forms a Precipitate
- $NaNO_3$: Remains Dissolved
- $BaSO_4$: Forms a Precipitate
- $K_2SO_4$: Remains Dissolved
- $PbI_2$: Forms a Precipitate