QUESTION IMAGE
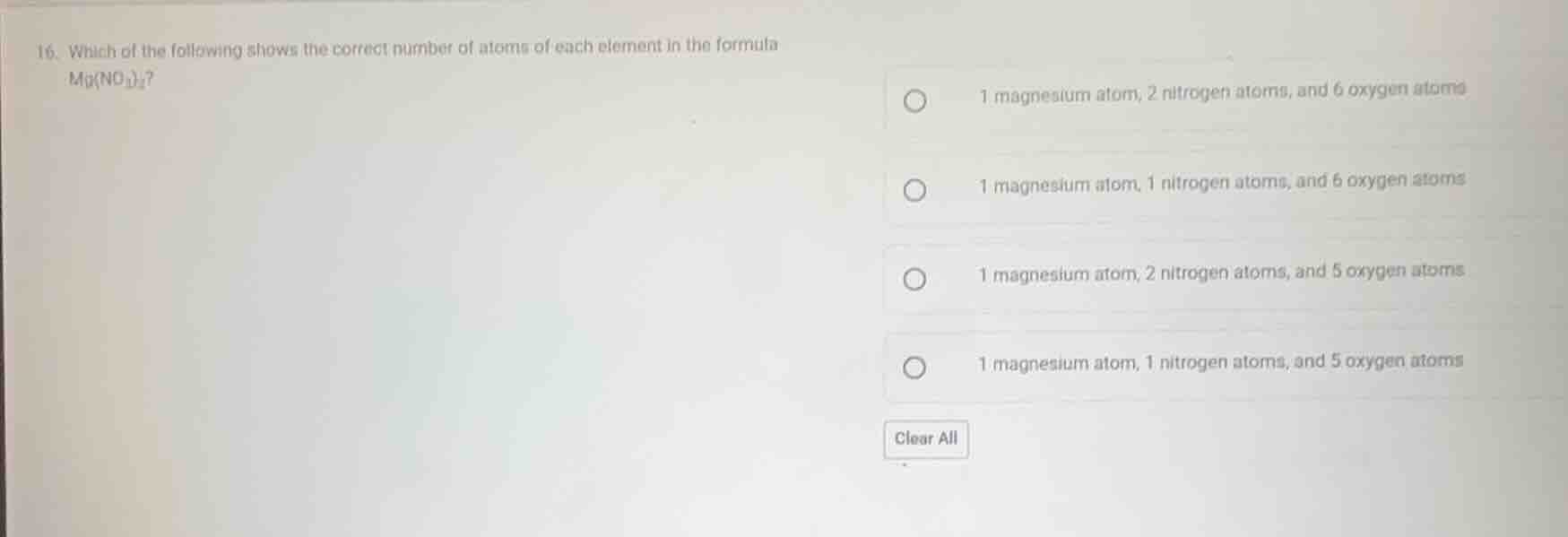
Question
- which of the following shows the correct number of atoms of each element in the formula mg(no₃)₂? 1 magnesium atom, 2 nitrogen atoms, and 6 oxygen atoms 1 magnesium atom, 1 nitrogen atoms, and 6 oxygen atoms 1 magnesium atom, 2 nitrogen atoms, and 5 oxygen atoms 1 magnesium atom, 1 nitrogen atoms, and 5 oxygen atoms clear all
Brief Explanations
To determine the number of atoms in \( \text{Mg(NO}_3\text{)}_2 \), we analyze each element:
- Magnesium (Mg): There is 1 Mg atom (no subscript or coefficient modifying it outside the parentheses).
- Nitrogen (N): Inside the parentheses, \( \text{NO}_3 \) has 1 N atom, and the subscript 2 outside the parentheses multiplies the entire \( \text{NO}_3 \) group. So, \( 1\times2 = 2 \) N atoms.
- Oxygen (O): Inside the parentheses, \( \text{NO}_3 \) has 3 O atoms, and the subscript 2 outside multiplies this. So, \( 3\times2 = 6 \) O atoms.
This matches the first option: 1 magnesium atom, 2 nitrogen atoms, and 6 oxygen atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1 magnesium atom, 2 nitrogen atoms, and 6 oxygen atoms (the first option)