QUESTION IMAGE
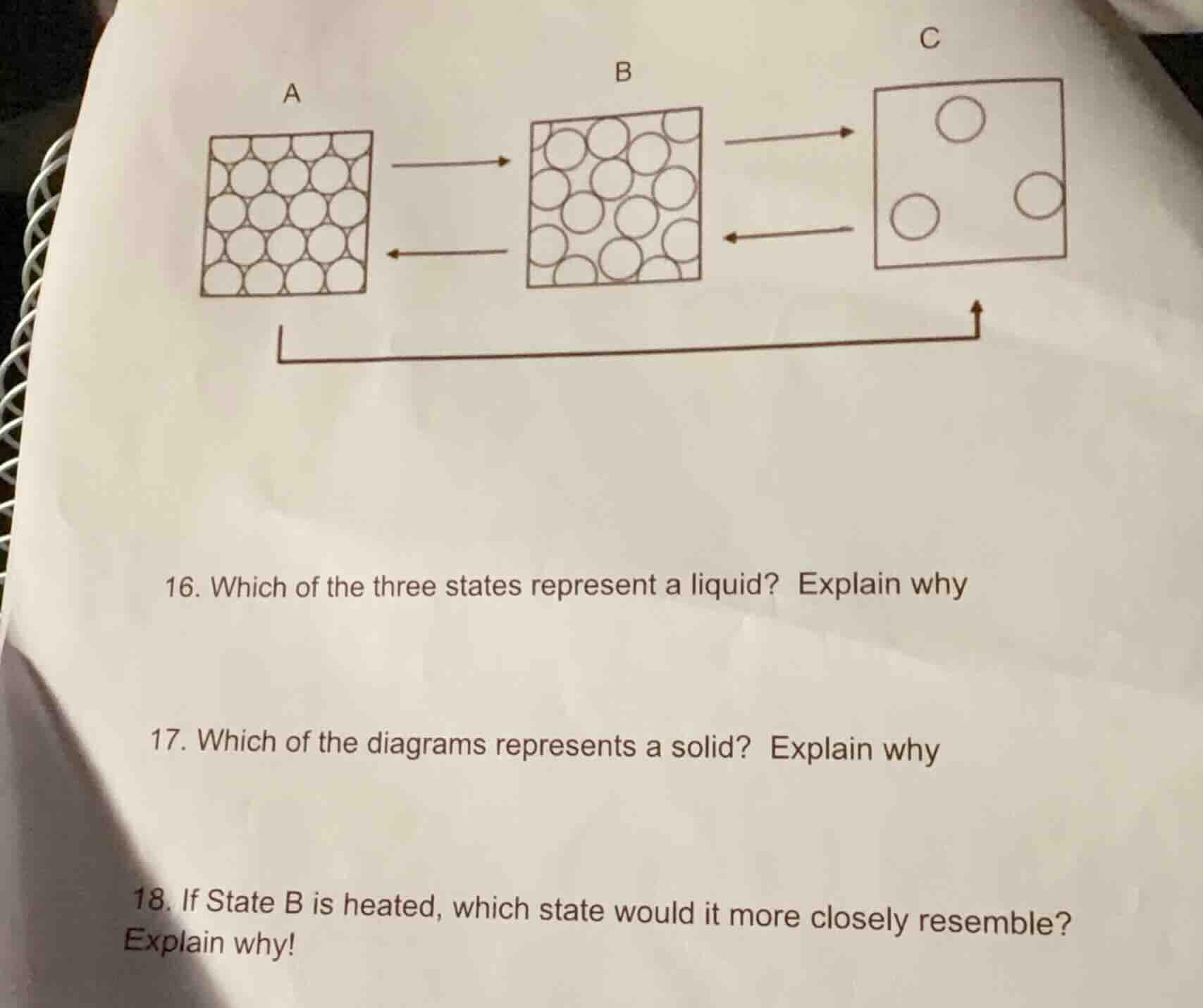
Question
- which of the three states represent a liquid? explain why
- which of the diagrams represents a solid? explain why
- if state b is heated, which state would it more closely resemble? explain why!
Question 16
In the particle model of matter, liquids have particles that are close together but can move past each other (not in a fixed, rigid arrangement like solids, nor far apart like gases). Diagram B shows particles that are relatively close but not in a strict, ordered pattern (unlike A, which looks like a solid with fixed positions) and not as spread out as C (which looks like a gas). So B represents a liquid because its particles are close but can move, fitting liquid's properties.
Solids have particles in a fixed, ordered, closely - packed arrangement (they vibrate in place but don't move freely). Diagram A shows particles in a regular, tightly - packed pattern, which is characteristic of a solid’s particle arrangement (unlike B’s more mobile - looking particles or C’s spread - out particles). So A represents a solid.
When a liquid (State B) is heated, it gains energy, and its particles move faster and spread out more, transitioning towards a gas. State C has particles that are far apart and move freely, which is the behavior of a gas. Heating State B (liquid) will cause its particles to gain energy, move more, and spread out, so it will resemble State C (gas) more closely.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B represents a liquid. Liquids have particles close (but not rigidly fixed like solids) and able to move past each other; B’s particles are close but not in a fixed pattern, matching liquid behavior.