QUESTION IMAGE
Question
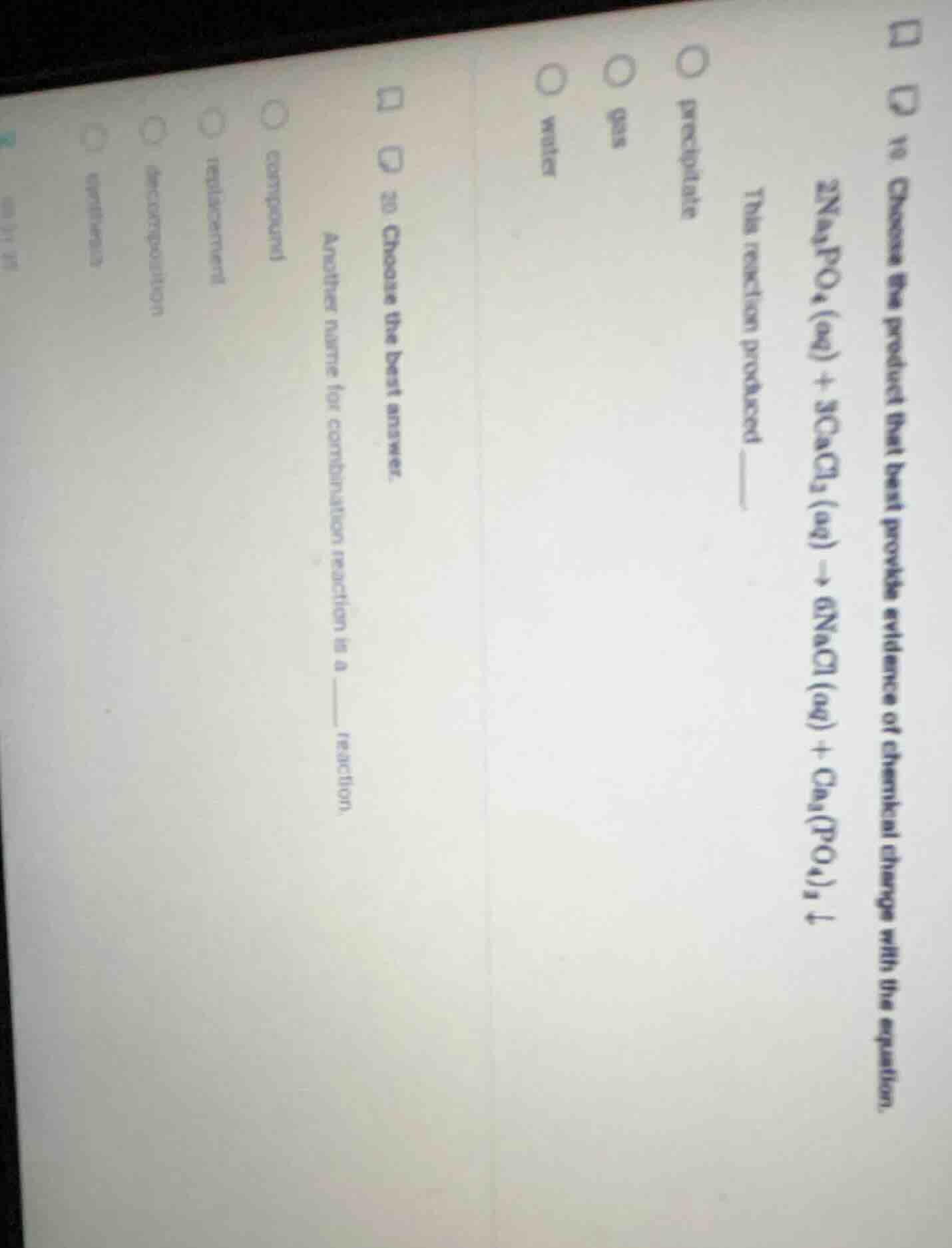
- choose the product that best provides evidence of chemical change with the equation. \\(2\text{na}_3\text{po}_4(\text{aq}) + 3\text{cacl}_2(\text{aq}) \
ightarrow 6\text{nacl}(\text{aq}) + \text{ca}_3(\text{po}_4)_2 \downarrow\\) this reaction produced ____ \\(\circ\\) precipitate \\(\circ\\) gas \\(\circ\\) water \\(\\) 20. choose the best answer. another name for combination reaction is a ____ reaction. \\(\circ\\) combustion \\(\circ\\) replacement \\(\circ\\) decomposition \\(\circ\\) synthesis
Step1: Analyze reaction 19
Look at the given equation: $2\text{Na}_3\text{PO}_4(aq) + 3\text{CaCl}_2(aq)
ightarrow 6\text{NaCl}(aq) + \text{Ca}_3(\text{PO}_4)_2 \downarrow$. The downward arrow ($\downarrow$) indicates a solid precipitate forms, which is visible evidence of chemical change.
Step2: Analyze reaction 20
A combination reaction (where reactants combine into one product) is also called a synthesis reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- precipitate
- synthesis