QUESTION IMAGE
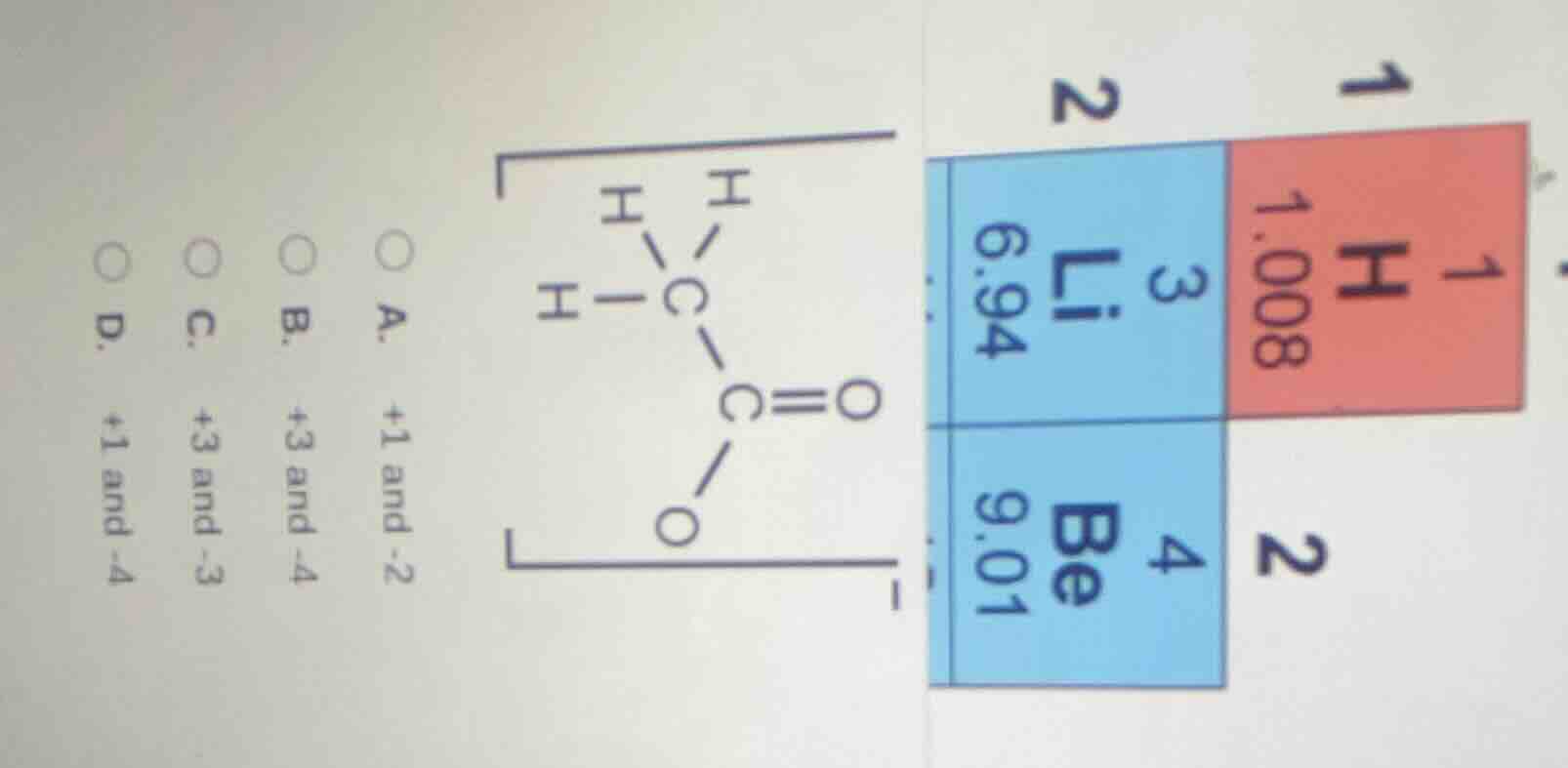
Question
a.
+1 and -2
b.
+3 and -4
c.
+3 and -3
d.
+1 and -4
Response
To solve this, we analyze the oxidation states of atoms in the compound (likely a carboxylate or related, with H, C, O).
Step 1: Oxidation State of H
Hydrogen (H) in most compounds (except hydrides) has an oxidation state of \( +1 \).
Step 2: Oxidation State of O
Oxygen (O) in most compounds (except peroxides/superoxides) has an oxidation state of \( -2 \).
Step 3: Analyze Carbon Atoms
- The first C (attached to 3 H and 1 C) and the second C (attached to C, O=, and O⁻) need balancing. But the key is matching oxidation states: H is \( +1 \), O is \( -2 \). The options involve \( +1 \) (H), \( -2 \) (O), and C’s states, but the question likely asks for H and O (or C, but the options suggest H and O).
Looking at the options, Option A is \( +1 \) (H) and \( -2 \) (O), which matches the typical oxidation states of H and O in organic/inorganic compounds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. +1 and -2