QUESTION IMAGE
Question
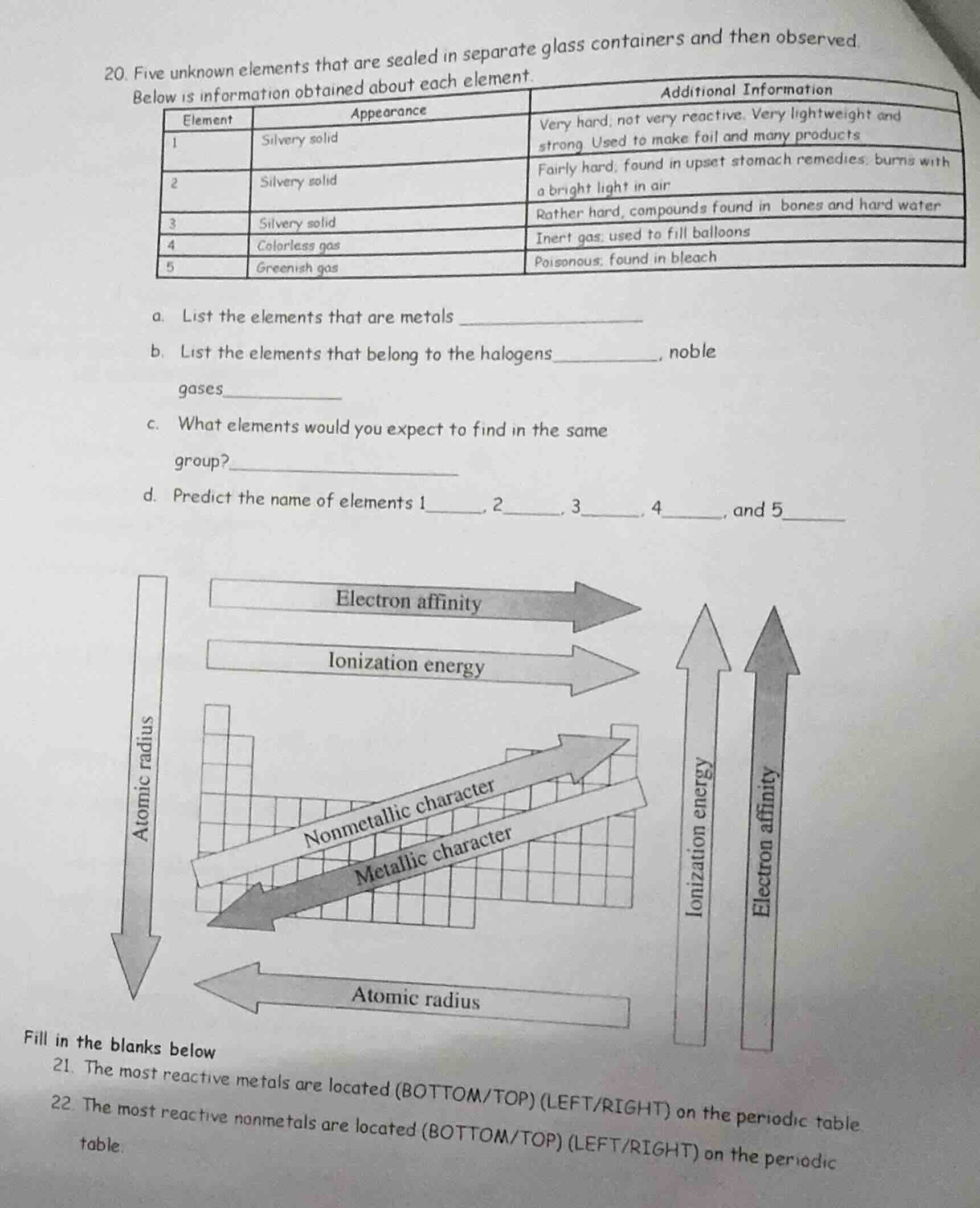
- five unknown elements that are sealed in separate glass containers and then observed. below is information obtained about each element.
| element | appearance | additional information |
|---|---|---|
| 2 | silvery solid | fairly hard, found in upset stomach remedies, burns with a bright light in air |
| 3 | silvery solid | rather hard, compounds found in bones and hard water |
| 4 | colorless gas | inert gas, used to fill balloons |
| 5 | greenish gas | poisonous, found in bleach |
a. list the elements that are metals ______________
b. list the elements that belong to the halogens_______, noble gases________
c. what elements would you expect to find in the same group?______________
d. predict the name of elements 1___, 2_, 3_, 4_, and 5___
chart about atomic radius, electron affinity, ionization energy, metallic character, nonmetallic character trends
fill in the blanks below
- the most reactive metals are located (bottom/top) (left/right) on the periodic table.
- the most reactive nonmetals are located (bottom/top) (left/right) on the periodic table.
For Question 20:
a. Metals are typically shiny solids (at standard conditions) with metallic properties. Elements 1,2,3 are silvery solids matching metal traits.
b. Halogens are reactive nonmetals; element 5 is a poisonous greenish gas in bleach (a halogen trait). Noble gases are inert; element 4 is an inert gas for balloons.
c. Elements 1,2,3 are all alkali/alkaline earth metals, so they share group properties.
d. Match traits to known elements:
- Lightweight, strong, silvery solid for foil = Aluminum
- Used in antacids, burns bright = Magnesium
- In bones/hard water = Calcium
- Inert gas for balloons = Helium
- Poisonous greenish gas in bleach = Chlorine
For Questions 21-22:
- Periodic table trend: Most reactive metals are at bottom-left (low ionization energy, high metallic character).
- Most reactive nonmetals are at top-right (high electron affinity/ionization energy, high nonmetallic character).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
20.
a. Elements 1, 2, 3
b. halogens: Element 5; noble gases: Element 4
c. Elements 1, 2, 3
d. 1: Aluminum; 2: Magnesium; 3: Calcium; 4: Helium; 5: Chlorine
- BOTTOM, LEFT
- TOP, RIGHT