QUESTION IMAGE
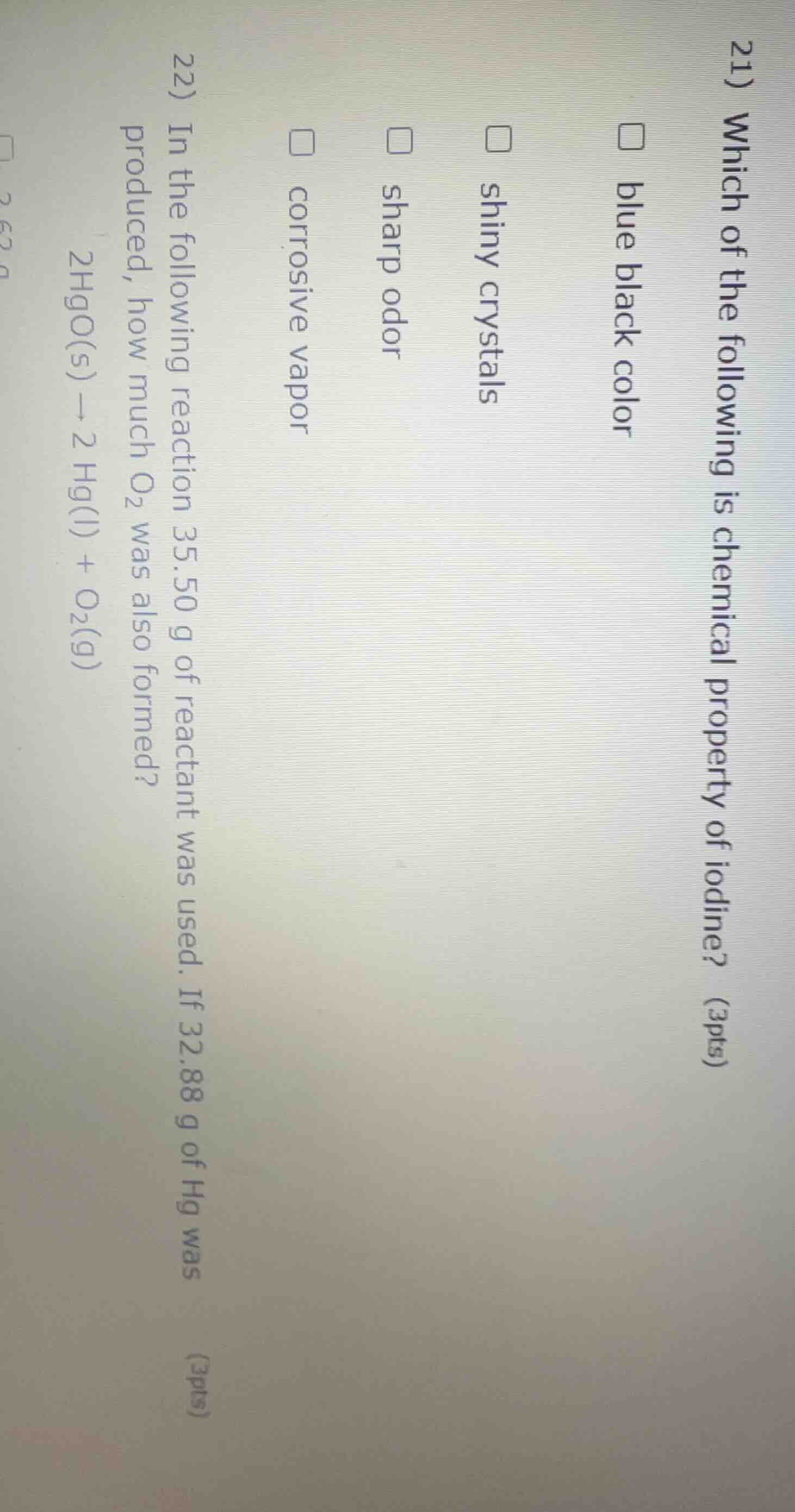
Question
- which of the following is chemical property of iodine? (3pts)
□ blue black color
□ shiny crystals
□ sharp odor
□ corrosive vapor
- in the following reaction 35.50 g of reactant was used. if 32.88 g of hg was produced, how much o₂ was also formed?
2hgo(s) → 2 hg(l) + o₂(g)
---
Question 21
Physical properties are observable without chemical change; chemical properties describe reactivity/behavior during chemical change. Blue-black color, shiny crystals, sharp odor are physical (observable without altering iodine's chemical structure). Corrosive vapor describes iodine's chemical reactivity (it damages materials via chemical reactions).
Step1: Apply Law of Conservation of Mass
The total mass of reactants equals total mass of products. Let $m_{\text{O}_2}$ = mass of oxygen.
$m_{\text{HgO}} = m_{\text{Hg}} + m_{\text{O}_2}$
Step2: Rearrange to solve for $m_{\text{O}_2}$
Substitute given masses into the equation.
$m_{\text{O}_2} = m_{\text{HgO}} - m_{\text{Hg}}$
$m_{\text{O}_2} = 35.50\ \text{g} - 32.88\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\square$ blue black color
$\square$ shiny crystals
$\square$ sharp odor
$\checkmark$ corrosive vapor
---