QUESTION IMAGE
Question
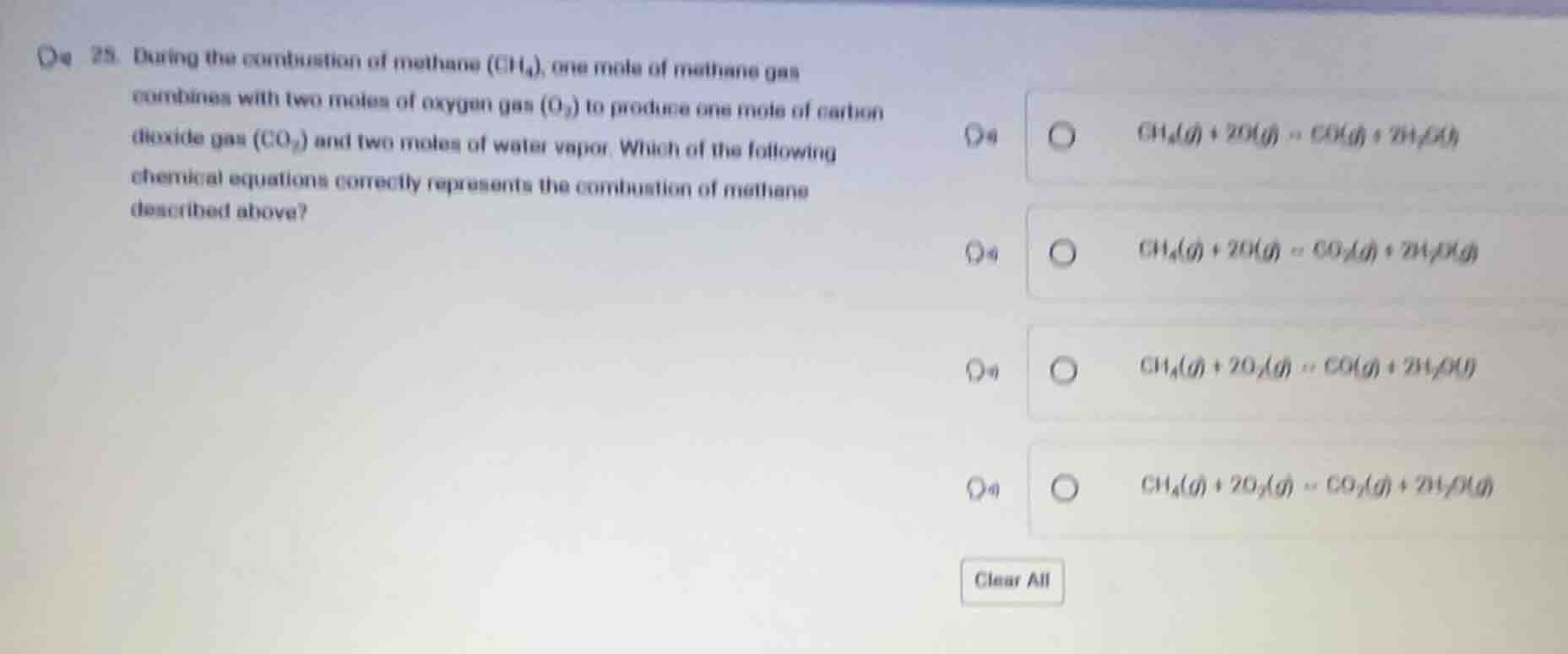
- during the combustion of methane (ch₄), one mole of methane gas combines with two moles of oxygen gas (o₂) to produce one mole of carbon dioxide gas (co₂) and two moles of water vapor. which of the following chemical equations correctly represents the combustion of methane described above? \\(\ce{ch_{4}(g) + 2o(g) = co_{2}(g) + 2h_{2}o(g)}\\) \\(\ce{ch_{4}(g) + 2o(g) = co_{2}(g) + 2h_{2}o(g)}\\) \\(\ce{ch_{4}(g) + 2o_{2}(g) = co(g) + 2h_{2}o(g)}\\) \\(\ce{ch_{4}(g) + 2o_{2}(g) = co_{2}(g) + 2h_{2}o(g)}\\)
Step1: Match reactant formulas
Reactants: $\text{CH}_4(g)$ and $2\text{O}_2(g)$
Step2: Match product formulas
Products: $\text{CO}_2(g)$ and $2\text{H}_2\text{O}(g)$ (water vapor = gas state)
Step3: Verify balanced equation
Check atom counts: C: 1=1, H: 4=4, O: 4=4, all balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\text{CH}_4(g) + 2\text{O}_2(g)
ightarrow \text{CO}_2(g) + 2\text{H}_2\text{O}(g)$