QUESTION IMAGE
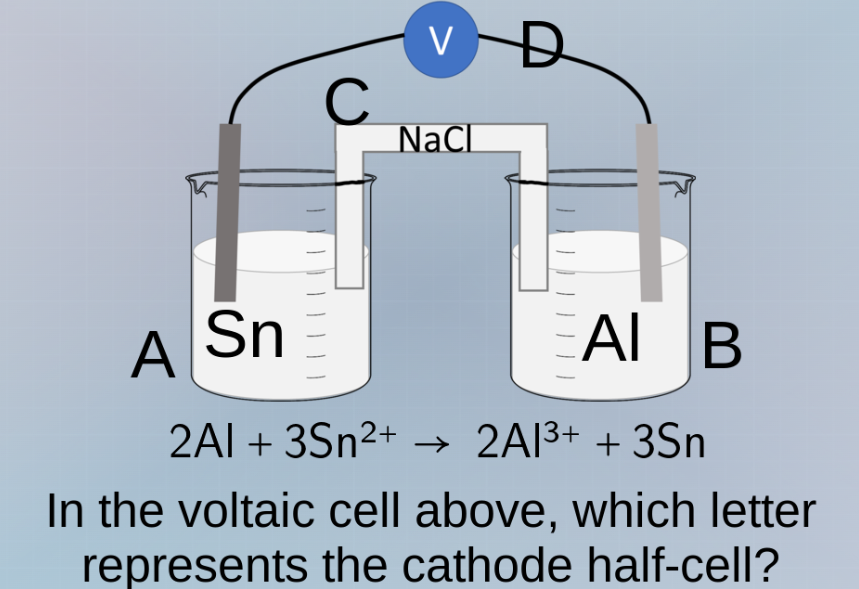
Question
2al + 3sn²⁺ → 2al³⁺ + 3sn
in the voltaic cell above, which letter represents the cathode half - cell?
In a voltaic cell, the cathode is where reduction occurs. The given reaction is \(2\text{Al} + 3\text{Sn}^{2+}
ightarrow2\text{Al}^{3+}+ 3\text{Sn}\). The reduction half - reaction is \(\text{Sn}^{2+}+2e^-
ightarrow\text{Sn}\) (since \(Sn^{2+}\) gains electrons to form Sn). The electrode where reduction takes place is the cathode. The Sn electrode is in beaker A. So the cathode half - cell is represented by letter A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A