QUESTION IMAGE
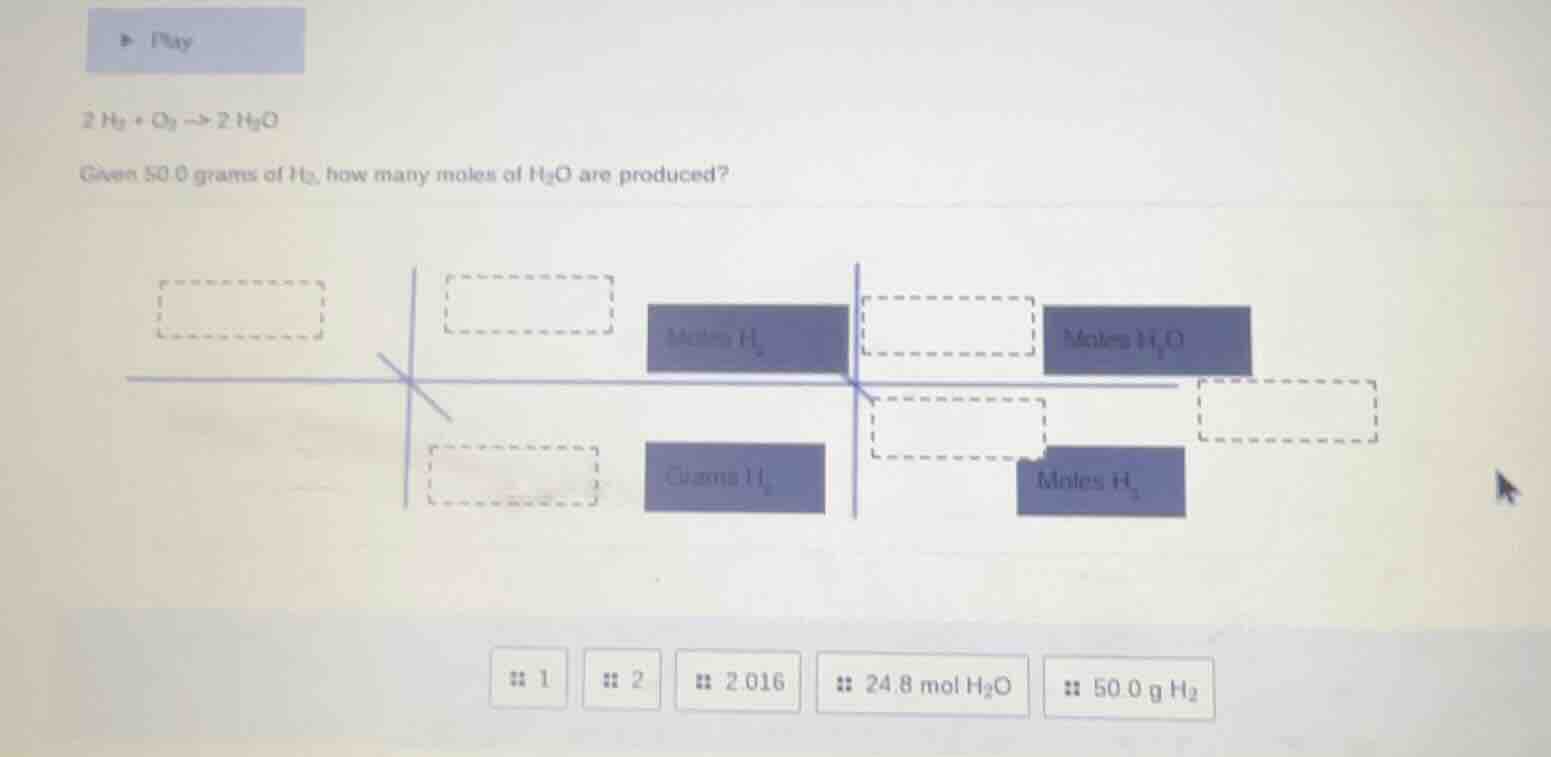
Question
$2h_2 + o_2 \
ightarrow 2h_2o$
given 50.0 grams of $h_2$, how many moles of $h_2o$ are produced?
:: 1
:: 2
:: 2.016
:: 24.8 mol $h_2o$
:: 50.0 g $h_2$
Step1: Identify given value
Given mass of $\text{H}_2$: $m_{\text{H}_2}=50.0\ \text{g}$
Step2: Molar mass of $\text{H}_2$
Molar mass $M_{\text{H}_2}=2.016\ \text{g/mol}$
Step3: Calculate moles of $\text{H}_2$
$$n_{\text{H}_2}=\frac{m_{\text{H}_2}}{M_{\text{H}_2}}=\frac{50.0\ \text{g}}{2.016\ \text{g/mol}}$$
Step4: Mole ratio from reaction
Reaction: $2\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}$, ratio $\frac{n_{\text{H}_2\text{O}}}{n_{\text{H}_2}}=\frac{2}{2}=1$
Step5: Calculate moles of $\text{H}_2\text{O}$
$$n_{\text{H}_2\text{O}}=n_{\text{H}_2} \times 1=\frac{50.0}{2.016}\ \text{mol} \approx 24.8\ \text{mol}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
24.8 mol $\text{H}_2\text{O}$
(For the box matching:
- Leftmost box: $\boldsymbol{50.0\ \text{g}\ \text{H}_2}$
- Middle left box: $\boldsymbol{2.016}$
- Middle right box upper: $\boldsymbol{1}$
- Middle right box lower: $\boldsymbol{2}$
- Rightmost box: $\boldsymbol{24.8\ \text{mol}\ \text{H}_2\text{O}}$)