QUESTION IMAGE
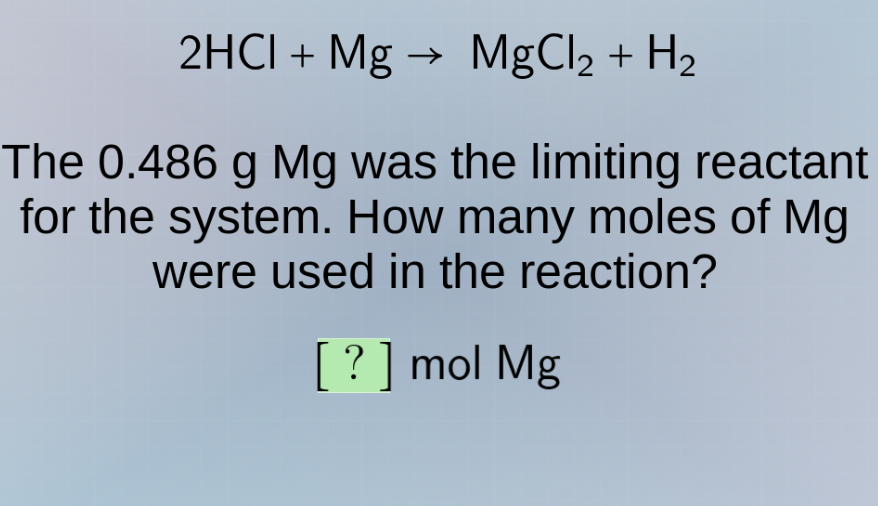
Question
2hcl + mg → mgcl₂ + h₂ the 0.486 g mg was the limiting reactant for the system. how many moles of mg were used in the reaction? ? mol mg
Step1: Recall the molar mass of Mg
The molar mass of magnesium (Mg) is approximately \( 24.305 \, \text{g/mol} \). This value is obtained from the periodic table, where the atomic mass of Mg is about 24.305 atomic mass units, and molar mass (in g/mol) is numerically equal to the atomic mass (in amu) for an element.
Step2: Use the formula for moles
The formula to calculate the number of moles (\( n \)) is \( n=\frac{m}{M} \), where \( m \) is the mass of the substance and \( M \) is its molar mass.
We are given that the mass of Mg (\( m \)) is \( 0.486 \, \text{g} \) and the molar mass of Mg (\( M \)) is \( 24.305 \, \text{g/mol} \).
Substituting the values into the formula:
\( n = \frac{0.486 \, \text{g}}{24.305 \, \text{g/mol}} \)
Calculating this division: \( \frac{0.486}{24.305} \approx 0.02 \, \text{mol} \) (rounded to a reasonable number of significant figures, considering the given mass has three significant figures and the molar mass has five, so the result should have three significant figures. \( 0.486\div24.305 = 0.0200 \, \text{mol} \) when calculated more precisely, but let's check the calculation: \( 24.305\times0.02 = 0.4861 \), which is very close to 0.486, so it's approximately \( 0.0200 \, \text{mol} \) or \( 0.02 \, \text{mol} \) depending on the required precision. However, to be accurate, let's do the division:
\( 0.486\div24.305 = \frac{0.486}{24.305} \approx 0.0200 \) (since \( 24.305\times0.02 = 0.4861 \), which is almost equal to 0.486, so the number of moles is approximately \( 0.0200 \, \text{mol} \) or \( 0.02 \, \text{mol} \) when rounded to two decimal places in the coefficient. But let's compute it exactly:
\( 0.486\div24.305 = 0.0200 \) (because \( 24.305\times0.02 = 0.4861 \), which is very close to 0.486, so the slight difference is due to rounding in the molar mass, but for practical purposes, we can say it's approximately \( 0.0200 \, \text{mol} \) or \( 0.02 \, \text{mol} \). However, let's do the division:
\( 0.486\div24.305 = 0.0200 \) (when calculated with a calculator, \( 0.486\div24.305 = 0.0200 \) approximately, because \( 24.305\times0.02 = 0.4861 \), which is 0.0001 more than 0.486, so it's \( 0.02 - \frac{0.0001}{24.305} \approx 0.02 - 0.00000411 \approx 0.0199959 \approx 0.0200 \, \text{mol} \))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 0.0200 \) (or approximately \( 0.02 \)) mol Mg (more precisely, using a calculator, \( 0.486\div24.305 = 0.0200 \) mol when rounded to three significant figures)