QUESTION IMAGE
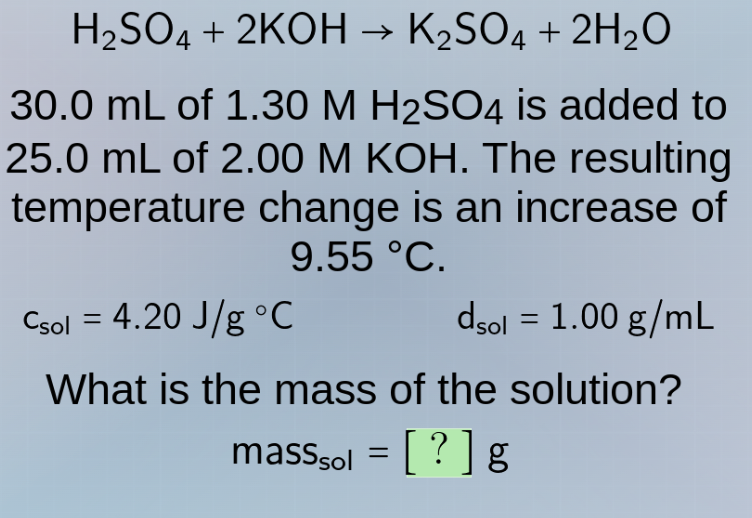
Question
h₂so₄ + 2koh → k₂so₄ + 2h₂o
30.0 ml of 1.30 m h₂so₄ is added to
25.0 ml of 2.00 m koh. the resulting
temperature change is an increase of
9.55 °c.
cₛₒₗ = 4.20 j/g °c dₛₒₗ = 1.00 g/ml
what is the mass of the solution?
massₛₒₗ = ? g
Step1: Find total volume of solution
The volume of \(H_2SO_4\) is \(30.0\) mL and the volume of \(KOH\) is \(25.0\) mL. To find the total volume of the solution, we add these two volumes together.
\[
V_{\text{total}} = 30.0\ \text{mL} + 25.0\ \text{mL} = 55.0\ \text{mL}
\]
Step2: Calculate mass of solution using density
The density of the solution (\(d_{\text{sol}}\)) is given as \(1.00\ \text{g/mL}\). The formula that relates mass (\(m\)), density (\(d\)), and volume (\(V\)) is \(m = d\times V\). We substitute the values of density and total volume into this formula.
\[
m_{\text{sol}} = d_{\text{sol}} \times V_{\text{total}} = 1.00\ \text{g/mL} \times 55.0\ \text{mL} = 55.0\ \text{g}
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(55.0\)