QUESTION IMAGE
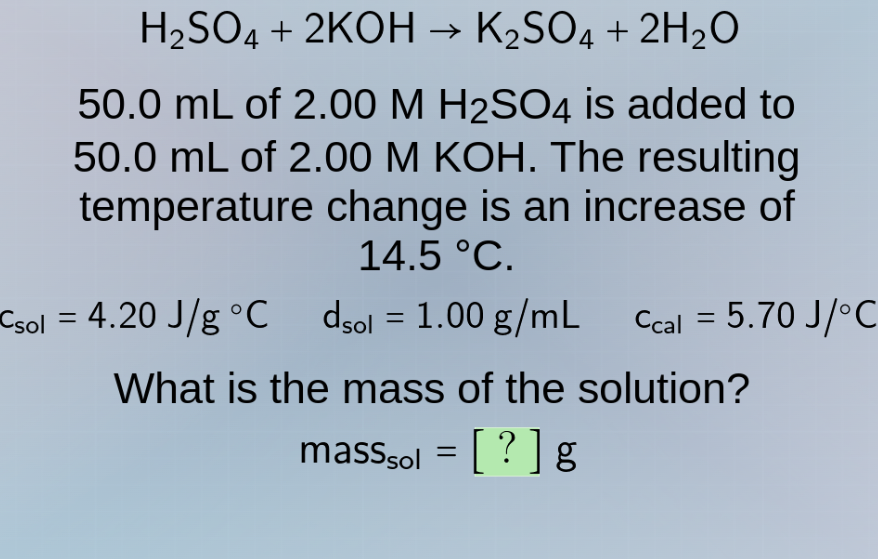
Question
h₂so₄ + 2koh → k₂so₄ + 2h₂o
50.0 ml of 2.00 m h₂so₄ is added to
50.0 ml of 2.00 m koh. the resulting
temperature change is an increase of
14.5 °c.
c_sol = 4.20 j/g °c d_sol = 1.00 g/ml c_cal = 5.70 j/°c
what is the mass of the solution?
mass_sol = ? g
Step1: Determine total volume of solution
The volume of \(H_2SO_4\) is \(50.0\space mL\) and the volume of \(KOH\) is \(50.0\space mL\). So the total volume \(V_{total}=50.0 + 50.0=100.0\space mL\).
Step2: Calculate mass of solution using density
The density of the solution \(d_{sol}=1.00\space g/mL\). The formula for mass is \(mass = density\times volume\). So \(mass_{sol}=d_{sol}\times V_{total}\). Substituting the values, we get \(mass_{sol}=1.00\space g/mL\times100.0\space mL\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
100.0