QUESTION IMAGE
Question
9/35
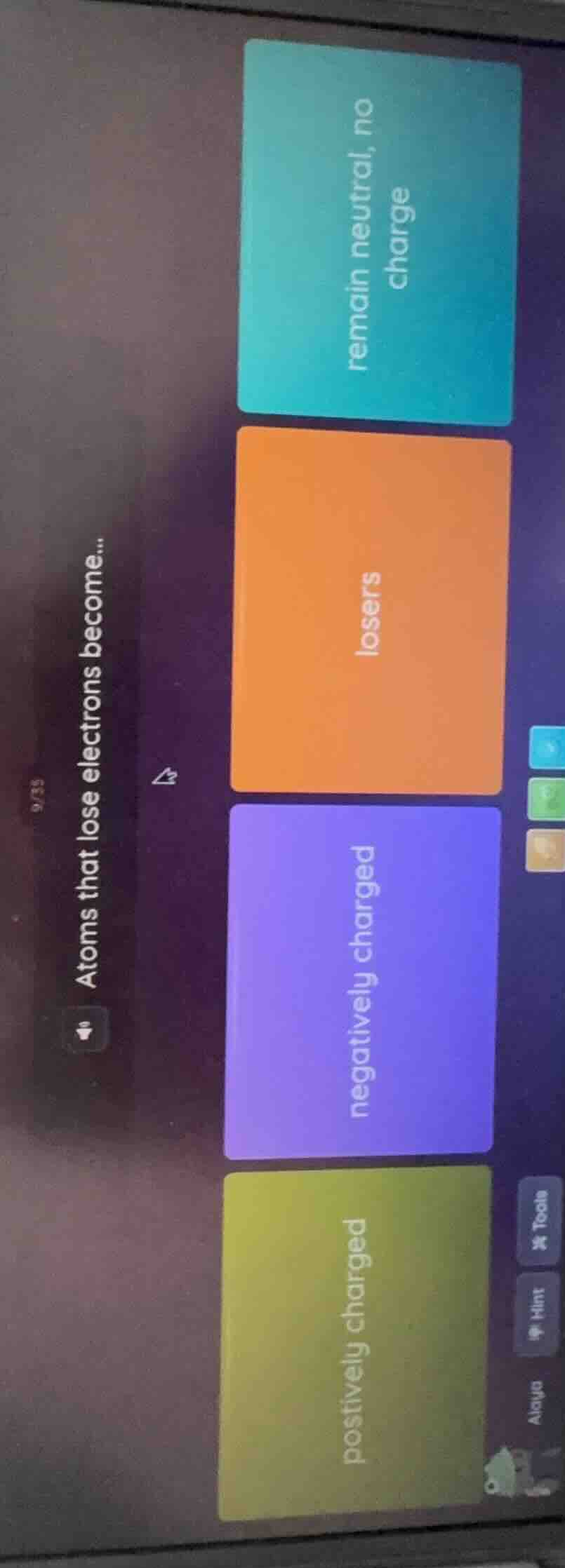
atoms that lose electrons become...
remain neutral, no charge
losers
negatively charged
postively charged
Brief Explanations
Atoms have equal numbers of positively charged protons and negatively charged electrons, making them neutral. When an atom loses electrons, it has more protons than electrons, resulting in a net positive charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
positively charged