QUESTION IMAGE
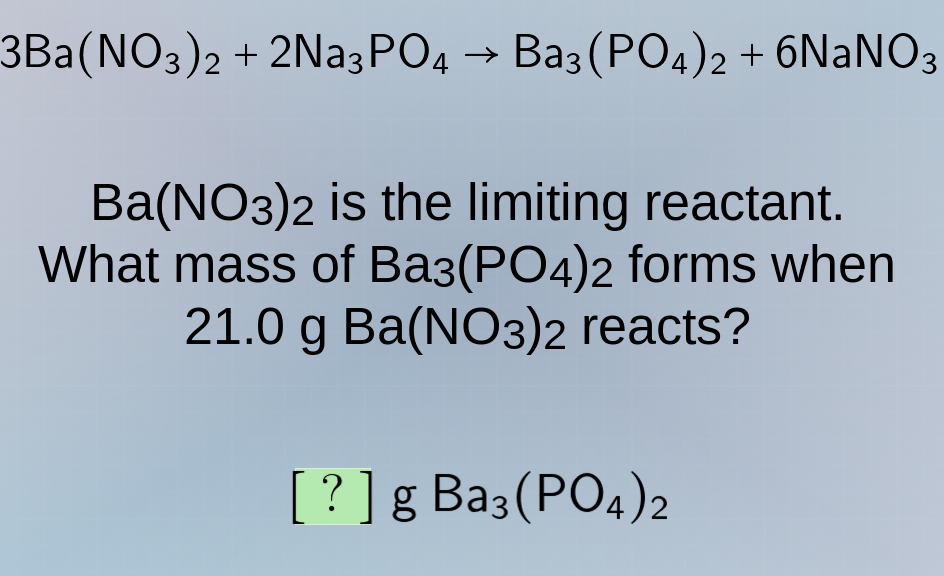
Question
3ba(no₃)₂ + 2na₃po₄ → ba₃(po₄)₂ + 6nano₃
ba(no₃)₂ is the limiting reactant.
what mass of ba₃(po₄)₂ forms when
21.0 g ba(no₃)₂ reacts?
? g ba₃(po₄)₂
Step1: Calculate moles of \( \text{Ba(NO}_3\text{)}_2 \)
Molar mass of \( \text{Ba(NO}_3\text{)}_2 \): \( \text{Ba} = 137.33 \, \text{g/mol} \), \( \text{N} = 14.01 \, \text{g/mol} \), \( \text{O} = 16.00 \, \text{g/mol} \).
Molar mass \( = 137.33 + 2\times(14.01 + 3\times16.00) = 261.34 \, \text{g/mol} \).
Moles \( = \frac{\text{mass}}{\text{molar mass}} = \frac{21.0 \, \text{g}}{261.34 \, \text{g/mol}} \approx 0.08036 \, \text{mol} \).
Step2: Use stoichiometry to find moles of \( \text{Ba}_3(\text{PO}_4)_2 \)
From the reaction: \( 3 \, \text{mol} \, \text{Ba(NO}_3\text{)}_2
ightarrow 1 \, \text{mol} \, \text{Ba}_3(\text{PO}_4)_2 \).
Moles of \( \text{Ba}_3(\text{PO}_4)_2 = \frac{0.08036 \, \text{mol}}{3} \approx 0.02679 \, \text{mol} \).
Step3: Calculate molar mass of \( \text{Ba}_3(\text{PO}_4)_2 \)
\( \text{Ba} = 137.33 \, \text{g/mol} \), \( \text{P} = 30.97 \, \text{g/mol} \), \( \text{O} = 16.00 \, \text{g/mol} \).
Molar mass \( = 3\times137.33 + 2\times(30.97 + 4\times16.00) = 601.93 \, \text{g/mol} \).
Step4: Calculate mass of \( \text{Ba}_3(\text{PO}_4)_2 \)
Mass \( = \text{moles} \times \text{molar mass} = 0.02679 \, \text{mol} \times 601.93 \, \text{g/mol} \approx 16.1 \, \text{g} \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \boxed{16.1} \)