QUESTION IMAGE
Question
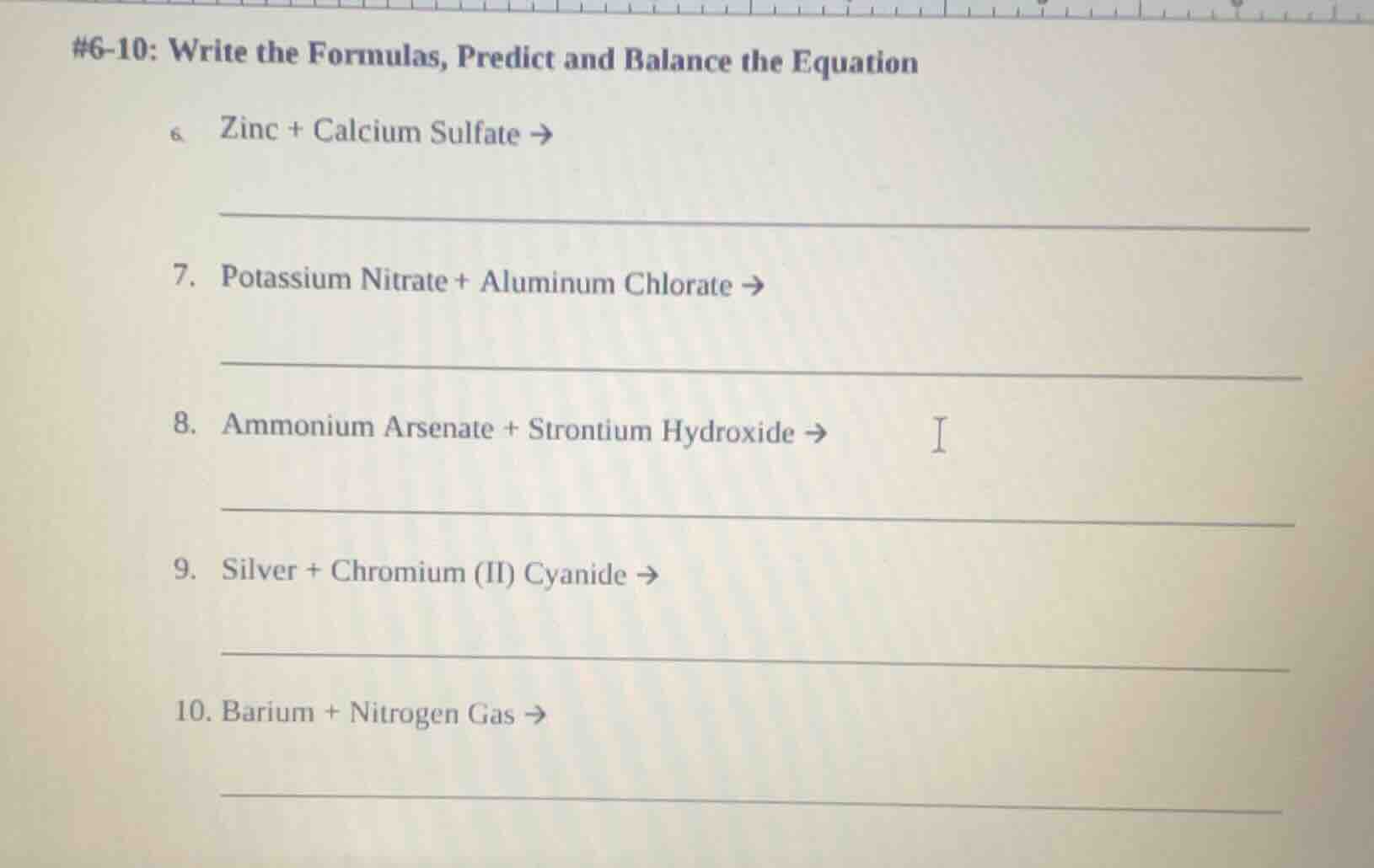
#6-10: write the formulas, predict and balance the equation
- zinc + calcium sulfate →
_________________________
- potassium nitrate + aluminum chlorate →
_________________________
- ammonium arsenate + strontium hydroxide →
_________________________
- silver + chromium (ii) cyanide →
_________________________
- barium + nitrogen gas →
_________________________
Problem 6: Zinc + Calcium Sulfate →
Step 1: Identify Reactants and Possible Reaction Type
Zinc (Zn) is a metal, Calcium Sulfate ($\text{CaSO}_4$) is a salt. Check if a single - replacement reaction occurs (Zn displaces Ca? Check activity series: Ca is more reactive than Zn, so no reaction.
Step 2: Write the Conclusion
Since Ca is more reactive than Zn, no chemical reaction takes place. So the equation is: $\text{Zn} + \text{CaSO}_4
ightarrow \text{No Reaction}$
Problem 7: Potassium Nitrate + Aluminum Chlorate →
Step 1: Identify Reactants and Reaction Type
Potassium Nitrate ($\text{KNO}_3$) and Aluminum Chlorate ($\text{Al(ClO}_3\text{)}_3$) are both salts. For a double - replacement reaction, we check if any precipitate, gas, or water is formed. The possible products are Potassium Chlorate ($\text{KClO}_3$) and Aluminum Nitrate ($\text{Al(NO}_3\text{)}_3$). Both are soluble in water.
Step 2: Write the Conclusion
Since no precipitate, gas, or water is formed, no reaction occurs. So the equation is: $\text{KNO}_3+\text{Al(ClO}_3\text{)}_3
ightarrow\text{No Reaction}$
Problem 8: Ammonium Arsenate + Strontium Hydroxide →
Step 1: Write Formulas of Reactants
Ammonium Arsenate: $(\text{NH}_4)_3\text{AsO}_4$, Strontium Hydroxide: $\text{Sr(OH)}_2$
Step 2: Predict Products (Double - Replacement)
Swap cations/anions: Ammonium Hydroxide ($\text{NH}_4\text{OH}$) and Strontium Arsenate ($\text{Sr}_3(\text{AsO}_4)_2$). $\text{NH}_4\text{OH}$ decomposes to $\text{NH}_3$ (gas) and $\text{H}_2\text{O}$ (liquid), and $\text{Sr}_3(\text{AsO}_4)_2$ is a precipitate (insoluble).
Step 3: Balance the Equation
First, write the unbalanced equation: $(\text{NH}_4)_3\text{AsO}_4+\text{Sr(OH)}_2
ightarrow\text{Sr}_3(\text{AsO}_4)_2+\text{NH}_3+\text{H}_2\text{O}$
Balance Sr: Put 3 in front of $\text{Sr(OH)}_2$: $(\text{NH}_4)_3\text{AsO}_4 + 3\text{Sr(OH)}_2
ightarrow\text{Sr}_3(\text{AsO}_4)_2+\text{NH}_3+\text{H}_2\text{O}$
Balance $\text{AsO}_4$: Put 2 in front of $(\text{NH}_4)_3\text{AsO}_4$: $2(\text{NH}_4)_3\text{AsO}_4+3\text{Sr(OH)}_2
ightarrow\text{Sr}_3(\text{AsO}_4)_2+\text{NH}_3+\text{H}_2\text{O}$
Balance $\text{NH}_4^+$ (which becomes $\text{NH}_3$): 2*3 = 6 $\text{NH}_4^+$, so put 6 in front of $\text{NH}_3$: $2(\text{NH}_4)_3\text{AsO}_4 + 3\text{Sr(OH)}_2
ightarrow\text{Sr}_3(\text{AsO}_4)_2+6\text{NH}_3+\text{H}_2\text{O}$
Balance O and H from $\text{OH}^-$ and $\text{H}_2\text{O}$: 32 = 6 $\text{OH}^-$, which gives 6 $\text{H}$ (from $\text{OH}^-$) and 6 $\text{O}$ (from $\text{OH}^-$). The $\text{NH}_4^+$ gives 6 $\text{H}$ (in $\text{NH}_3$ and $\text{H}_2\text{O}$). The number of O in $\text{Sr}_3(\text{AsO}_4)_2$: 24 = 8, in $2(\text{NH}_4)_3\text{AsO}_4$: 24 = 8, in $3\text{Sr(OH)}_2$: 32 = 6, in $6\text{NH}_3$: 0, in $\text{H}_2\text{O}$: let's say $x$. So 8 + 6=8 + 0+x*1→x = 6. So put 6 in front of $\text{H}_2\text{O}$.
The balanced equation: $2(\text{NH}_4)_3\text{AsO}_4+3\text{Sr(OH)}_2=\text{Sr}_3(\text{AsO}_4)_2\downarrow+6\text{NH}_3\uparrow+6\text{H}_2\text{O}$
Problem 9: Silver + Chromium (II) Cyanide →
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- $\boldsymbol{\text{Zn} + \text{CaSO}_4
ightarrow\text{No Reaction}}$
- $\boldsymbol{\text{KNO}_3+\text{Al(ClO}_3\text{)}_3
ightarrow\text{No Reaction}}$
- $\boldsymbol{2(\text{NH}_4)_3\text{AsO}_4 + 3\text{Sr(OH)}_2=\text{Sr}_3(\text{AsO}_4)_2\downarrow+6\text{NH}_3\uparrow+6\text{H}_2\text{O}}$
- $\boldsymbol{\text{Ag}+\text{Cr(CN)}_2
ightarrow\text{No Reaction}}$
- $\boldsymbol{3\text{Ba}+\text{N}_2=\text{Ba}_3\text{N}_2}$