QUESTION IMAGE
Question
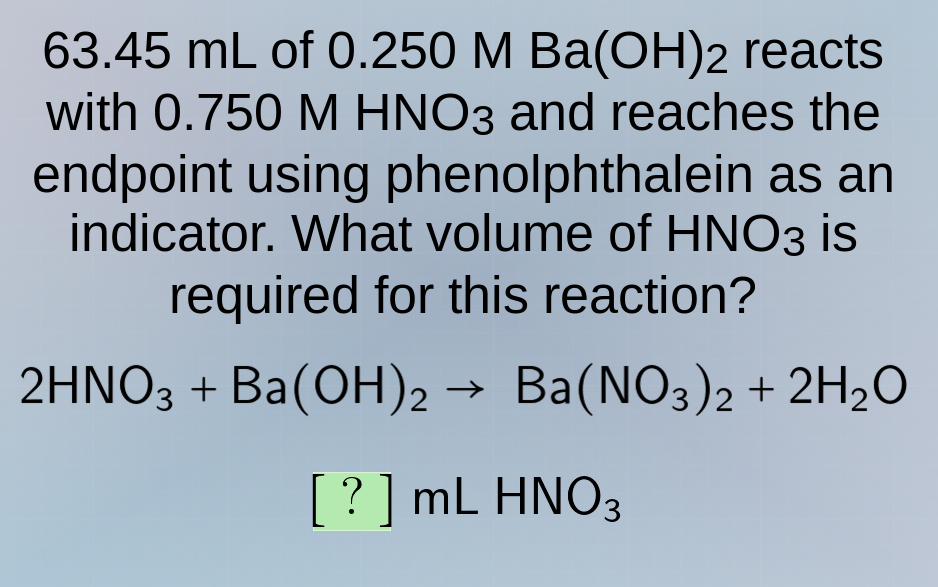
63.45 ml of 0.250 m ba(oh)₂ reacts with 0.750 m hno₃ and reaches the endpoint using phenolphthalein as an indicator. what volume of hno₃ is required for this reaction?
2hno₃ + ba(oh)₂ → ba(no₃)₂ + 2h₂o
? ml hno₃
Step1: Find moles of \( Ba(OH)_2 \)
Molarity \( M = \frac{n}{V} \), so \( n = M \times V \). Convert volume to liters: \( V = 63.45 \, mL = 0.06345 \, L \).
\( n_{Ba(OH)_2} = 0.250 \, M \times 0.06345 \, L = 0.0158625 \, mol \).
Step2: Use stoichiometry to find moles of \( HNO_3 \)
From the reaction: \( 2 \, mol \, HNO_3 \) reacts with \( 1 \, mol \, Ba(OH)_2 \).
\( n_{HNO_3} = 2 \times n_{Ba(OH)_2} = 2 \times 0.0158625 \, mol = 0.031725 \, mol \).
Step3: Find volume of \( HNO_3 \)
Rearrange \( M = \frac{n}{V} \) to \( V = \frac{n}{M} \).
\( V_{HNO_3} = \frac{0.031725 \, mol}{0.750 \, M} = 0.0423 \, L \). Convert to mL: \( 0.0423 \, L \times 1000 = 42.3 \, mL \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
42.3