QUESTION IMAGE
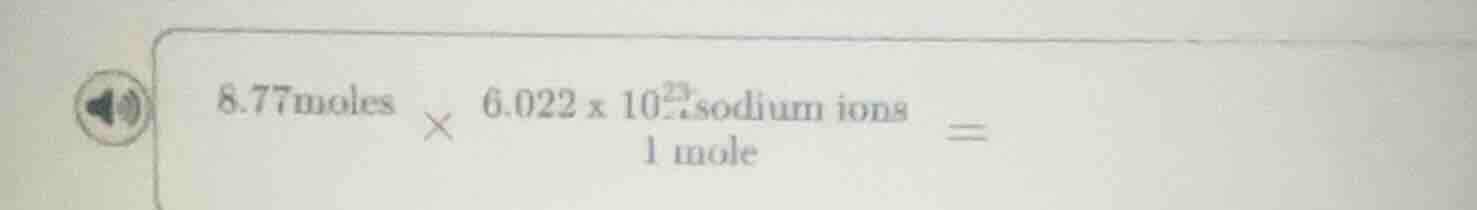
Question
8.77 moles × (6.022 × 10²³ sodium ions) / 1 mole =
Step1: Identify the operation
We need to multiply 8.77 moles by the conversion factor \(\frac{6.022\times10^{23}\text{ sodium ions}}{1\text{ mole}}\). The moles unit will cancel out, leaving us with the number of sodium ions.
Step2: Perform the multiplication
Calculate \(8.77\times6.022\times10^{23}\). First, multiply 8.77 and 6.022: \(8.77\times6.022 = 52.81394\). Then, multiply by \(10^{23}\): \(52.81394\times10^{23}=5.281394\times10^{24}\) (in scientific notation, we adjust the decimal to have one non - zero digit before the decimal point).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(5.28\times10^{24}\) (or \(5.281394\times10^{24}\)) sodium ions (the answer can be rounded to an appropriate number of significant figures, here 8.77 has three significant figures and 6.022 has four, so we can present it as \(5.28\times10^{24}\) or keep more decimal places as \(5.281\times10^{24}\) etc.)