QUESTION IMAGE
Question
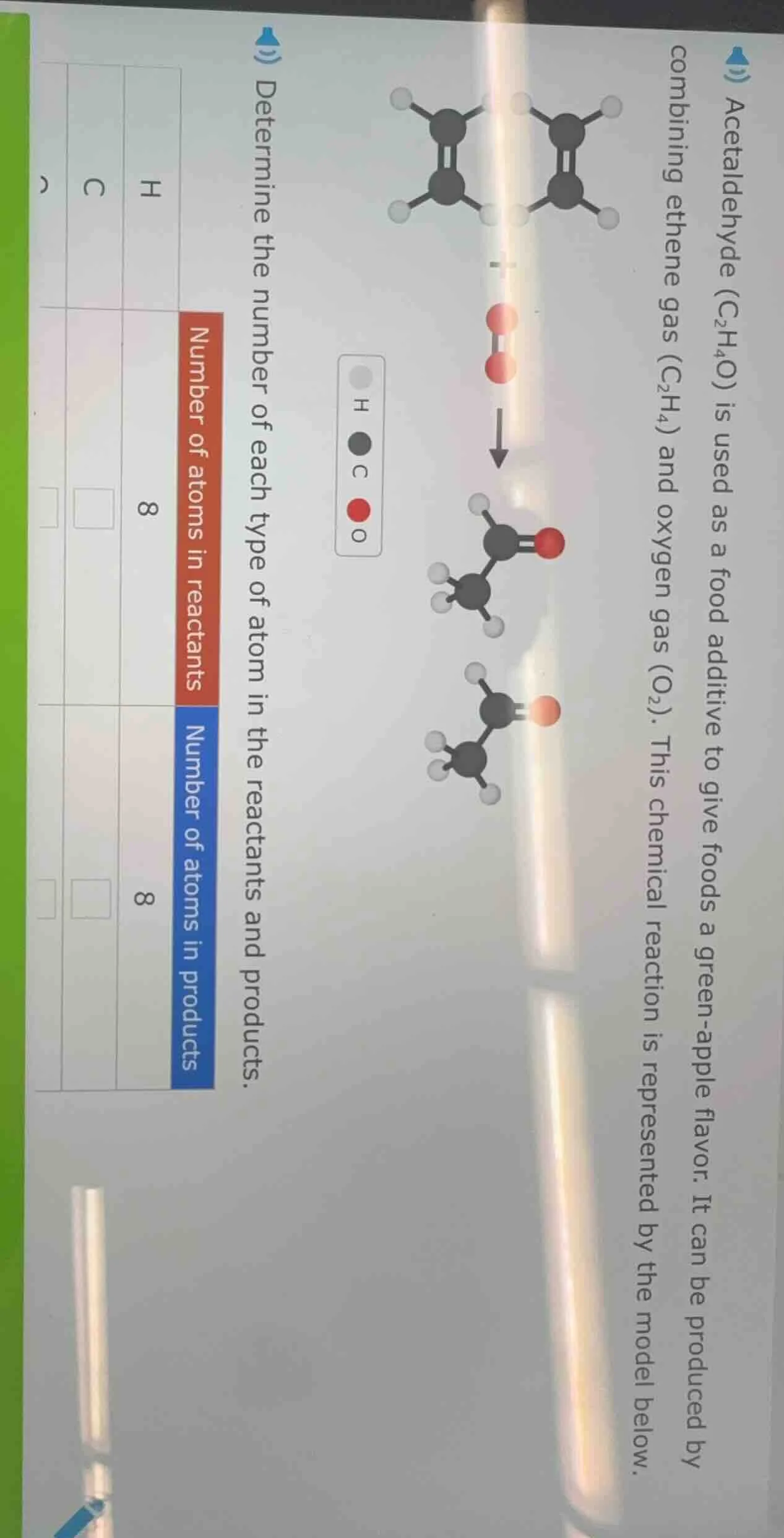
acetaldehyde ($c_2h_4o$) is used as a food additive to give foods a green-apple flavor. it can be produced by combining ethene gas ($c_2h_4$) and oxygen gas ($o_2$). this chemical reaction is represented by the model below.determine the number of each type of atom in the reactants and products.| | number of atoms in reactants | number of atoms in products || ---- | ---- | ---- || h | 8 | 8 || c | | || o | | |
Step1: Count H atoms in reactants
Each $\text{C}_2\text{H}_4$ has 4 H; 2 molecules: $2 \times 4 = 8$
Step2: Count C atoms in reactants
Each $\text{C}_2\text{H}_4$ has 2 C; 2 molecules: $2 \times 2 = 4$
Step3: Count O atoms in reactants
$\text{O}_2$ has 2 O; 1 molecule: $1 \times 2 = 2$
Step4: Count H atoms in products
Each $\text{C}_2\text{H}_4\text{O}$ has 4 H; 2 molecules: $2 \times 4 = 8$
Step5: Count C atoms in products
Each $\text{C}_2\text{H}_4\text{O}$ has 2 C; 2 molecules: $2 \times 2 = 4$
Step6: Count O atoms in products
Each $\text{C}_2\text{H}_4\text{O}$ has 1 O; 2 molecules: $2 \times 1 = 2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Atom | Number of atoms in reactants | Number of atoms in products |
|---|---|---|
| C | 4 | 4 |
| O | 2 | 2 |