QUESTION IMAGE
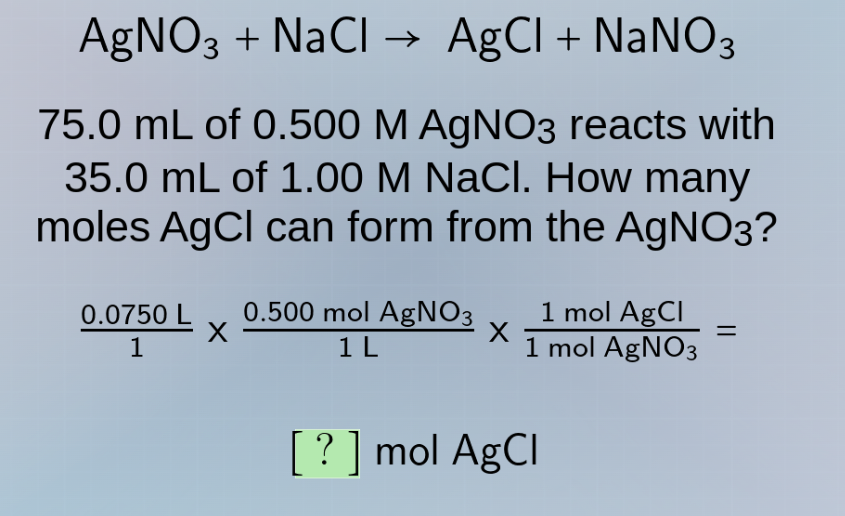
Question
agno₃ + nacl → agcl + nano₃
75.0 ml of 0.500 m agno₃ reacts with
35.0 ml of 1.00 m nacl. how many
moles agcl can form from the agno₃?
(0.0750 l / 1) × (0.500 mol agno₃ / 1 l) × (1 mol agcl / 1 mol agno₃) =
? mol agcl
Step1: Convert volume to liters
The volume of \(AgNO_3\) is 75.0 mL, which is \(0.0750\) L (since \(1\) L = \(1000\) mL, so \(75.0\div1000 = 0.0750\) L).
Step2: Calculate moles of \(AgNO_3\)
Using the molarity formula \(n = M\times V\) (where \(n\) is moles, \(M\) is molarity, \(V\) is volume in liters), for \(AgNO_3\) with \(M = 0.500\) M and \(V = 0.0750\) L, the moles of \(AgNO_3\) is \(0.0750\space L\times\frac{0.500\space mol\space AgNO_3}{1\space L}\).
Step3: Use stoichiometry to find moles of \(AgCl\)
From the balanced equation, the mole ratio of \(AgCl\) to \(AgNO_3\) is \(1:1\), so we multiply the moles of \(AgNO_3\) by \(\frac{1\space mol\space AgCl}{1\space mol\space AgNO_3}\).
Now, calculating the product:
\[
0.0750\times0.500\times1 = 0.0375
\]
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(0.0375\)