QUESTION IMAGE
Question
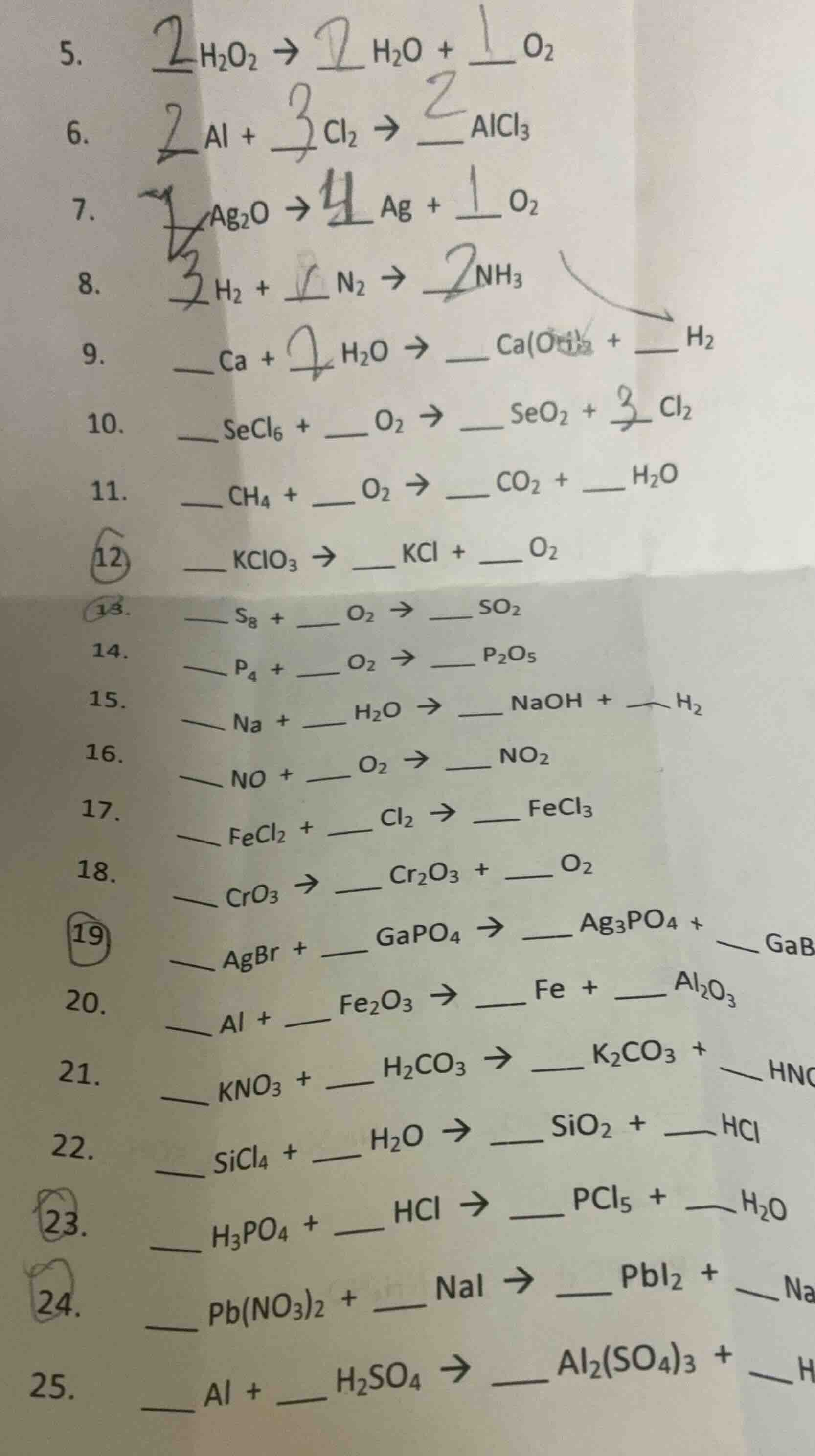
- h₂o₂ → h₂o + __o₂
- al + cl₂ → __alcl₃
- ag₂o → ag + __o₂
- h₂ + n₂ → __nh₃
- ca + h₂o → ca(oh)₂ + h₂
- secl₆ + o₂ → seo₂ + cl₂
- ch₄ + o₂ → co₂ + h₂o
- kclo₃ → kcl + __o₂
- s₈ + o₂ → __so₂
- p₄ + o₂ → __p₂o₅
- na + h₂o → naoh + h₂
- no + o₂ → __no₂
- fecl₂ + cl₂ → __fecl₃
- cro₃ → cr₂o₃ + __o₂
- agbr + gapo₄ → ag₃po₄ + gabr₃
- al + fe₂o₃ → fe + al₂o₃
- kno₃ + h₂co₃ → k₂co₃ + hno₃
- sicl₄ + h₂o → sio₂ + hcl
- h₃po₄ + hcl → pcl₅ + h₂o
- pb(no₃)₂ + nai → pbi₂ + nano₃
- al + h₂so₄ → al₂(so₄)₃ + h₂
Step1: Balance equation 9: Ca, H, O
Balance Ca first: $1\text{Ca} + \text{__H}_2\text{O}
ightarrow 1\text{Ca(OH)}_2 + \text{__H}_2$
Balance O: $1\text{Ca} + 2\text{H}_2\text{O}
ightarrow 1\text{Ca(OH)}_2 + \text{__H}_2$
Balance H: $1\text{Ca} + 2\text{H}_2\text{O}
ightarrow 1\text{Ca(OH)}_2 + 1\text{H}_2$
Step2: Balance equation10: Se, Cl, O
Balance Se first: $1\text{SeCl}_6 + \text{__O}_2
ightarrow 1\text{SeO}_2 + \text{__Cl}_2$
Balance Cl: $1\text{SeCl}_6 + \text{__O}_2
ightarrow 1\text{SeO}_2 + 3\text{Cl}_2$
Balance O: $1\text{SeCl}_6 + 1\text{__O}_2
ightarrow 1\text{SeO}_2 + 3\text{Cl}_2$
Step3: Balance equation11: C, H, O
Balance C first: $1\text{CH}_4 + \text{__O}_2
ightarrow 1\text{CO}_2 + \text{__H}_2\text{O}$
Balance H: $1\text{CH}_4 + \text{__O}_2
ightarrow 1\text{CO}_2 + 2\text{H}_2\text{O}$
Balance O: $1\text{CH}_4 + 2\text{O}_2
ightarrow 1\text{CO}_2 + 2\text{H}_2\text{O}$
Step4: Balance equation12: K, Cl, O
Balance K/Cl first: $2\text{KClO}_3
ightarrow 2\text{KCl} + \text{__O}_2$
Balance O: $2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2$
Step5: Balance equation13: S, O
Balance S first: $1\text{S}_8 + \text{__O}_2
ightarrow 8\text{SO}_2$
Balance O: $1\text{S}_8 + 8\text{O}_2
ightarrow 8\text{SO}_2$
Step6: Balance equation14: P, O
Balance P first: $1\text{P}_4 + \text{__O}_2
ightarrow 2\text{P}_2\text{O}_5$
Balance O: $1\text{P}_4 + 5\text{O}_2
ightarrow 2\text{P}_2\text{O}_5$
Step7: Balance equation15: Na, H, O
Balance Na first: $2\text{Na} + \text{__H}_2\text{O}
ightarrow 2\text{NaOH} + \text{__H}_2$
Balance H/O: $2\text{Na} + 2\text{H}_2\text{O}
ightarrow 2\text{NaOH} + 1\text{H}_2$
Step8: Balance equation16: N, O
Balance N first: $2\text{NO} + \text{__O}_2
ightarrow 2\text{NO}_2$
Balance O: $2\text{NO} + 1\text{O}_2
ightarrow 2\text{NO}_2$
Step9: Balance equation17: Fe, Cl
Balance Fe first: $2\text{FeCl}_2 + \text{__Cl}_2
ightarrow 2\text{FeCl}_3$
Balance Cl: $2\text{FeCl}_2 + 1\text{Cl}_2
ightarrow 2\text{FeCl}_3$
Step10: Balance equation18: Cr, O
Balance Cr first: $4\text{CrO}_3
ightarrow 2\text{Cr}_2\text{O}_3 + \text{__O}_2$
Balance O: $4\text{CrO}_3
ightarrow 2\text{Cr}_2\text{O}_3 + 3\text{O}_2$
Step11: Balance equation19: Ag, Ga, PO₄, Br
Balance Ag/PO₄ first: $3\text{AgBr} + 1\text{GaPO}_4
ightarrow 1\text{Ag}_3\text{PO}_4 + \text{__GaBr}_3$
Balance Ga/Br: $3\text{AgBr} + 1\text{GaPO}_4
ightarrow 1\text{Ag}_3\text{PO}_4 + 1\text{GaBr}_3$
Step12: Balance equation20: Al, Fe, O
Balance Al/Fe first: $2\text{Al} + 1\text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + 1\text{Al}_2\text{O}_3$
Balance O: $2\text{Al} + 1\text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + 1\text{Al}_2\text{O}_3$
Step13: Balance equation21: K, NO₃, CO₃, H
Balance K first: $2\text{KNO}_3 + \text{__H}_2\text{CO}_3
ightarrow 1\text{K}_2\text{CO}_3 + \text{__HNO}_3$
Balance NO₃/CO₃: $2\text{KNO}_3 + 1\text{H}_2\text{CO}_3
ightarrow 1\text{K}_2\text{CO}_3 + 2\text{HNO}_3$
Step14: Balance equation22: Si, Cl, H, O
Balance Si first: $1\text{SiCl}_4 + \text{__H}_2\text{O}
ightarrow 1\text{SiO}_2 + \text{__HCl}$
Balance Cl: $1\text{SiCl}_4 + \text{__H}_2\text{O}
ightarrow 1\text{SiO}_2 + 4\text{HCl}$
Balance H/O: $1\text{SiCl}_4 + 2\text{H}_2\text{O}
ightarrow 1\text{SiO}_2 + 4\text{HCl}$
Step15: Balance equation23: P, Cl, H, O
Balance P first: $1\text{H}_3\text{PO}_4 + \text{__HCl}
ightarrow 1\text{PCl}_5 + \text{__H}_2\text{O}$
Balance Cl: $1\text{H}_3\text{PO}_4 + 5\text{HCl}
ightarrow 1\text{PCl}_5 + \text{__H}_2\text{O}$
Balance H/O: $1\text{H}_3\text{PO}_4 + 5\text{HCl}
ightarrow 1\text{PCl}_5…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{1\text{Ca} + 2\text{H}_2\text{O}
ightarrow 1\text{Ca(OH)}_2 + 1\text{H}_2}$
- $\boldsymbol{1\text{SeCl}_6 + 1\text{O}_2
ightarrow 1\text{SeO}_2 + 3\text{Cl}_2}$
- $\boldsymbol{1\text{CH}_4 + 2\text{O}_2
ightarrow 1\text{CO}_2 + 2\text{H}_2\text{O}}$
- $\boldsymbol{2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2}$
- $\boldsymbol{1\text{S}_8 + 8\text{O}_2
ightarrow 8\text{SO}_2}$
- $\boldsymbol{1\text{P}_4 + 5\text{O}_2
ightarrow 2\text{P}_2\text{O}_5}$
- $\boldsymbol{2\text{Na} + 2\text{H}_2\text{O}
ightarrow 2\text{NaOH} + 1\text{H}_2}$
- $\boldsymbol{2\text{NO} + 1\text{O}_2
ightarrow 2\text{NO}_2}$
- $\boldsymbol{2\text{FeCl}_2 + 1\text{Cl}_2
ightarrow 2\text{FeCl}_3}$
- $\boldsymbol{4\text{CrO}_3
ightarrow 2\text{Cr}_2\text{O}_3 + 3\text{O}_2}$
- $\boldsymbol{3\text{AgBr} + 1\text{GaPO}_4
ightarrow 1\text{Ag}_3\text{PO}_4 + 1\text{GaBr}_3}$
- $\boldsymbol{2\text{Al} + 1\text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + 1\text{Al}_2\text{O}_3}$
- $\boldsymbol{2\text{KNO}_3 + 1\text{H}_2\text{CO}_3
ightarrow 1\text{K}_2\text{CO}_3 + 2\text{HNO}_3}$
- $\boldsymbol{1\text{SiCl}_4 + 2\text{H}_2\text{O}
ightarrow 1\text{SiO}_2 + 4\text{HCl}}$
- $\boldsymbol{1\text{H}_3\text{PO}_4 + 5\text{HCl}
ightarrow 1\text{PCl}_5 + 4\text{H}_2\text{O}}$
- $\boldsymbol{1\text{Pb(NO}_3\text{)}_2 + 2\text{NaI}
ightarrow 1\text{PbI}_2 + 2\text{NaNO}_3}$
- $\boldsymbol{2\text{Al} + 3\text{H}_2\text{SO}_4
ightarrow 1\text{Al}_2(\text{SO}_4\text{)}_3 + 3\text{H}_2}$
(Verified balanced coefficients for pre-filled questions 5-8:
- $\boldsymbol{2\text{H}_2\text{O}_2
ightarrow 2\text{H}_2\text{O} + 1\text{O}_2}$ (correct)
- $\boldsymbol{2\text{Al} + 3\text{Cl}_2
ightarrow 2\text{AlCl}_3}$ (correct)
- $\boldsymbol{2\text{Ag}_2\text{O}
ightarrow 4\text{Ag} + 1\text{O}_2}$ (correct)
- $\boldsymbol{3\text{H}_2 + 1\text{N}_2
ightarrow 2\text{NH}_3}$ (correct))