QUESTION IMAGE
Question
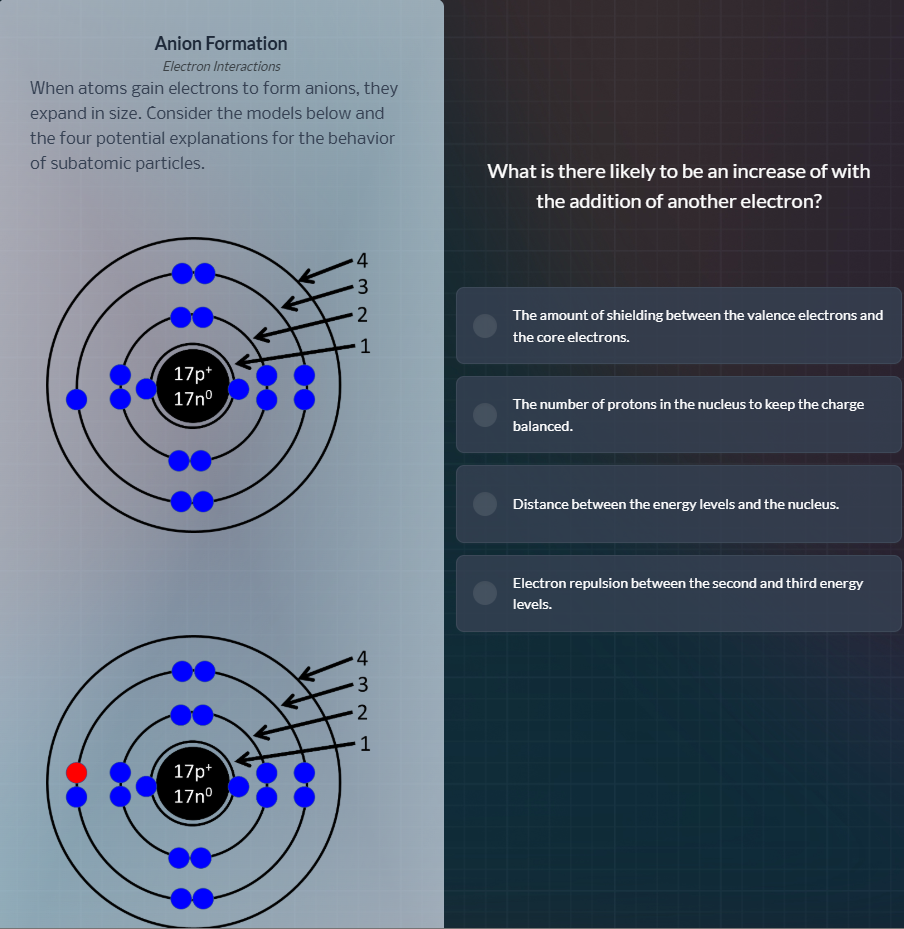
anion formation
electron interactions
when atoms gain electrons to form anions, they
expand in size. consider the models below and
the four potential explanations for the behavior
of subatomic particles.
what is there likely to be an increase of with
the addition of another electron?
the amount of shielding between the valence electrons and
the core electrons.
the number of protons in the nucleus to keep the charge
balanced.
distance between the energy levels and the nucleus.
electron repulsion between the second and third energy
levels.
When an atom gains an electron to form an anion, the proton count in the nucleus stays the same, so charge balance via protons does not occur. Shielding between core and valence electrons is determined by core electrons, which don't increase here. The expansion of the atom means energy levels move further from the nucleus, but the key direct effect of adding an electron is increased repulsion among electrons. In this case, the added electron is in the third energy level, increasing repulsion between electrons in the second and third energy levels.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Electron repulsion between the second and third energy levels.