QUESTION IMAGE
Question
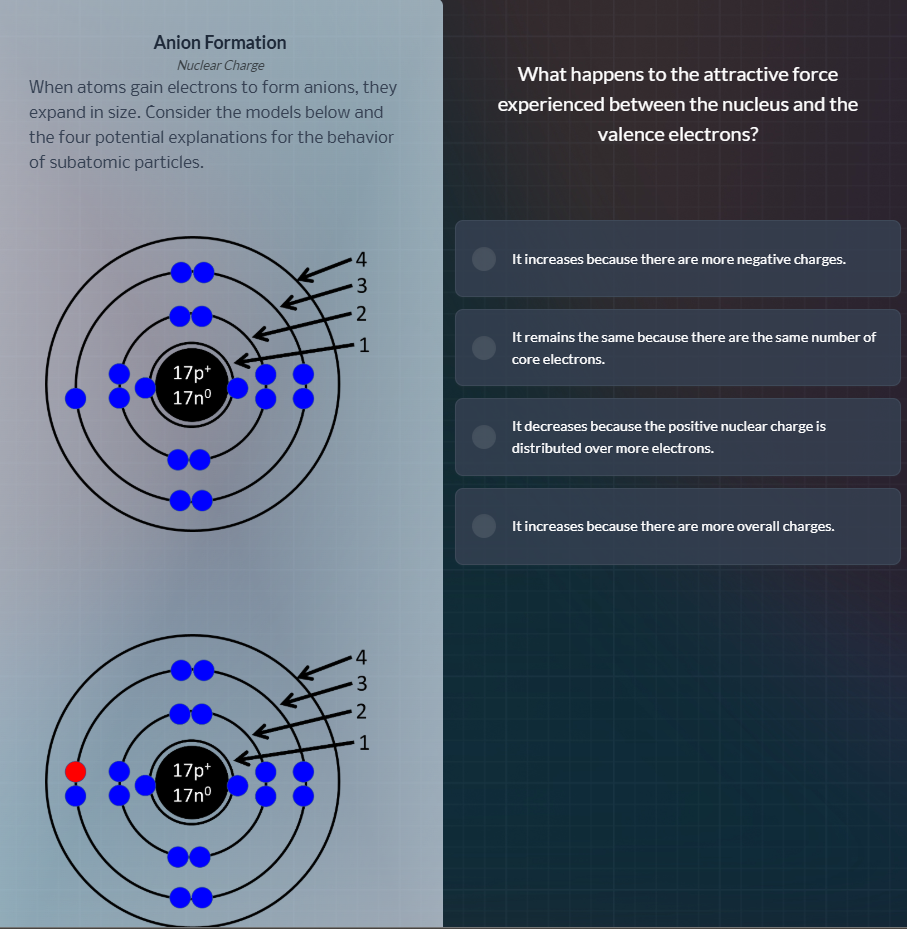
anion formation
nuclear charge
when atoms gain electrons to form anions, they
expand in size. consider the models below and
the four potential explanations for the behavior
of subatomic particles.
what happens to the attractive force
experienced between the nucleus and the
valence electrons?
it increases because there are more negative charges.
it remains the same because there are the same number of
core electrons.
it decreases because the positive nuclear charge is
distributed over more electrons.
it increases because there are more overall charges.
When forming an anion, the nucleus has a fixed positive charge (17 protons here) but gains additional valence electrons. The positive nuclear charge is now spread across more total electrons, so the attractive force per valence electron weakens, causing the electron cloud to expand.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
It decreases because the positive nuclear charge is distributed over more electrons.