QUESTION IMAGE
Question
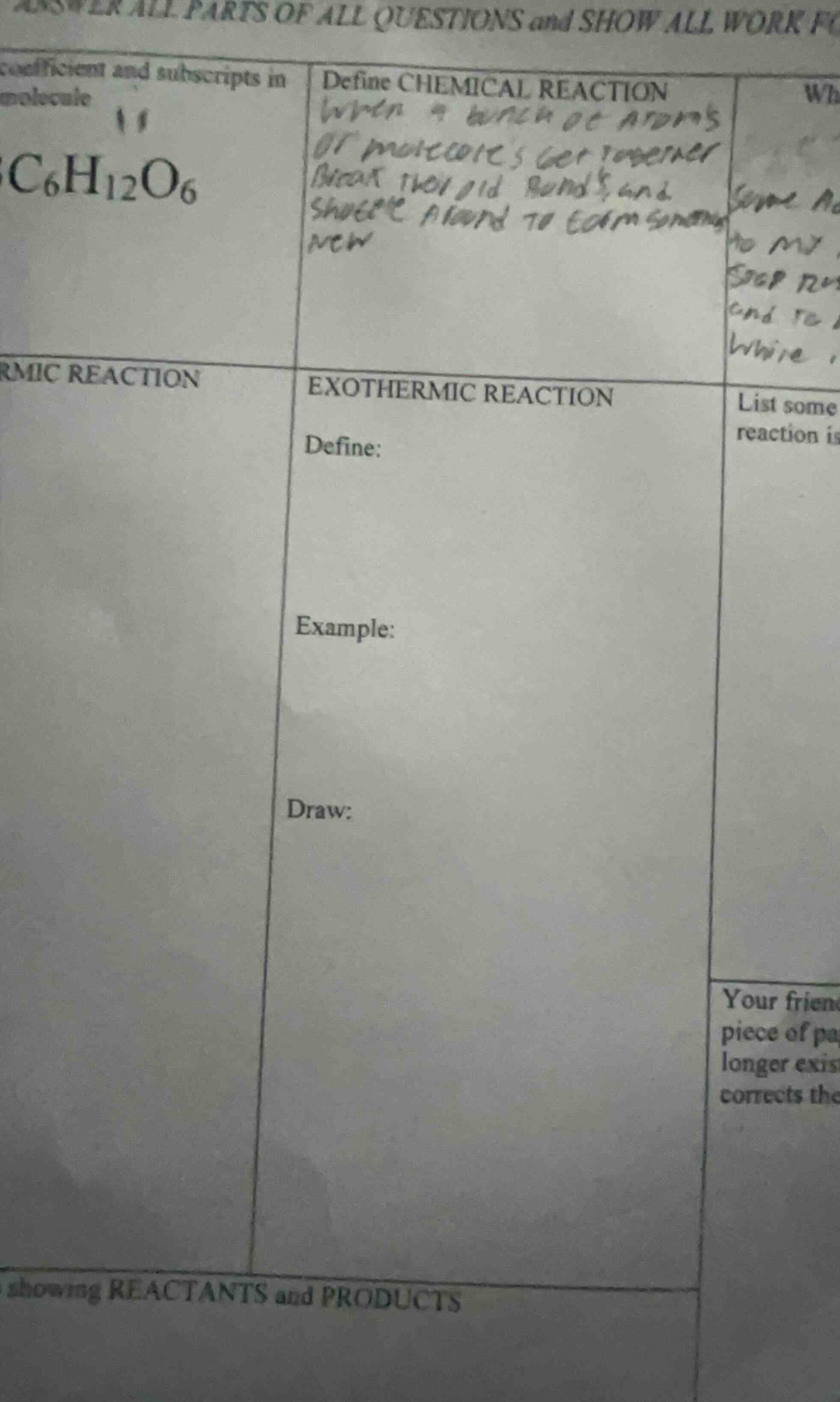
answer all parts of all questions and show all work ftext cut offcoefficient and subscripts in molecule11$\text{c}_6\text{h}_{12}\text{o}_6$define chemical reactionhandwritten text: when a bunch of atoms or molecules get together break their old bonds, and share a bond to form some newpartial text: whsome adto mystop wand towrire rmic reactionexothermic reactiondefine:example:draw:list somereaction isyour friendpiece of palonger existcorrects theshowing reactants and products
Brief Explanations
- Exothermic Reaction Definition: A chemical reaction that releases energy (usually as heat, light, or sound) into the surroundings, with the total enthalpy of products being less than that of reactants.
- Example: Combustion of methane is a common exothermic reaction, where methane reacts with oxygen to release heat, carbon dioxide, and water.
- Energy Diagram (Draw): The diagram has reactants at a higher energy level than products. A downward curve connects them, with a small peak (activation energy) between; label reactants, products, activation energy, and the energy released ($\Delta H < 0$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Define: A reaction that releases energy (heat/light) to the surroundings, where $\Delta H = H_{products} - H_{reactants} < 0$.
- Example: $\ce{CH4(g) + 2O2(g) -> CO2(g) + 2H2O(l) + Heat}$ (Combustion of methane)
- Draw:
- Y-axis: Energy, X-axis: Reaction Progress
- Plot reactants ($\ce{CH4 + 2O2}$) at a high energy point
- Draw a small upward peak (activation energy barrier)
- Plot products ($\ce{CO2 + 2H2O}$) at a lower energy point
- Add an arrow from reactants to products labeled "Energy Released ($\Delta H < 0$)"