QUESTION IMAGE
Question
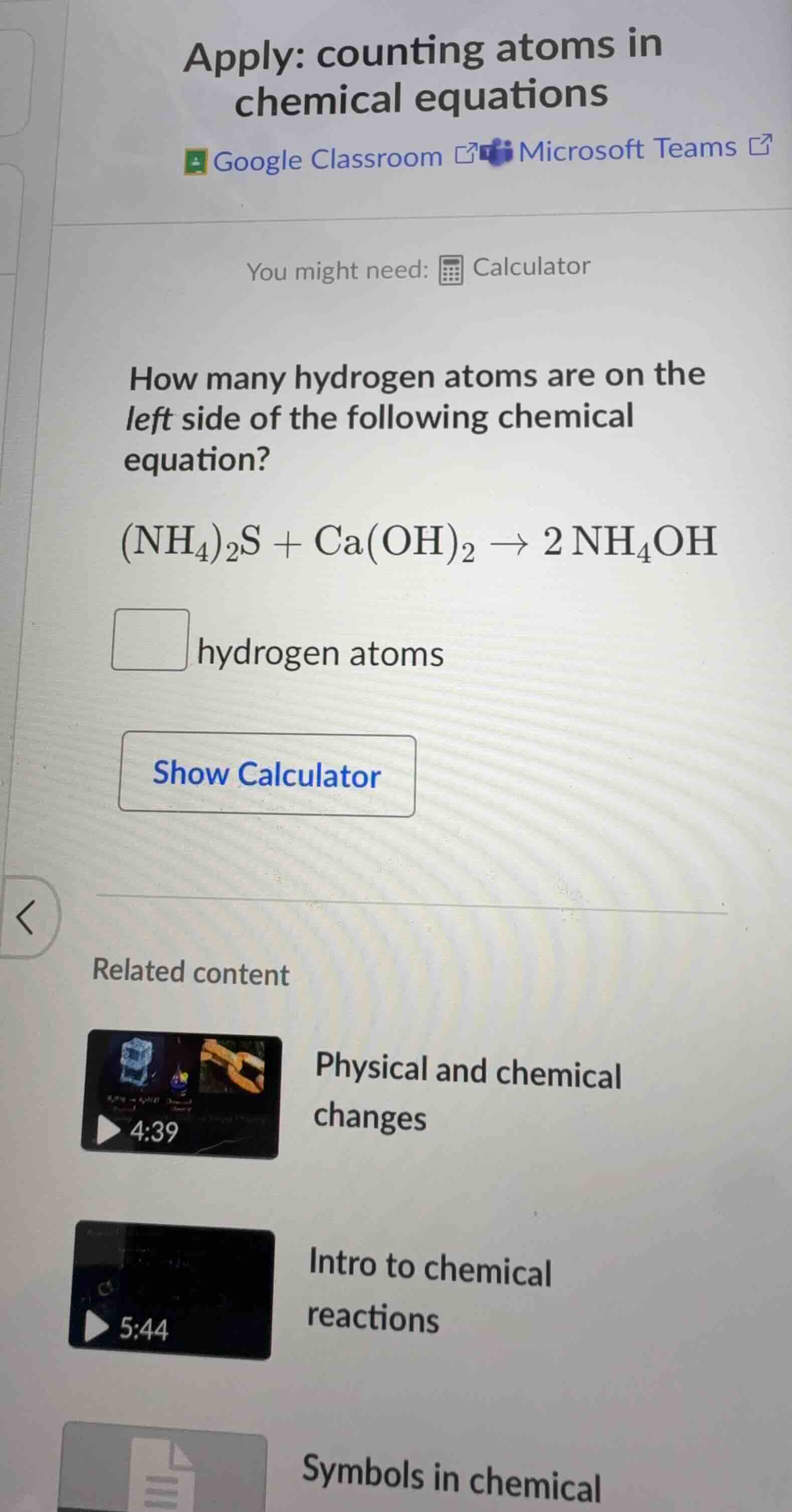
apply: counting atoms in chemical equations google classroom microsoft teams you might need: calculator how many hydrogen atoms are on the left side of the following chemical equation? \\((\text{nh}_4)_2\text{s} + \text{ca(oh)}_2 \
ightarrow 2\\ \text{nh}_4\text{oh}\\) hydrogen atoms show calculator related content physical and chemical changes intro to chemical reactions symbols in chemical
Step1: Analyze \((NH_4)_2S\)
In \((NH_4)_2S\), the subscript of \(NH_4\) is 2, and each \(NH_4\) has 4 H atoms. So H atoms from \((NH_4)_2S\): \(2\times4 = 8\).
Step2: Analyze \(Ca(OH)_2\)
In \(Ca(OH)_2\), each \(OH\) has 1 H atom, and there are 2 \(OH\) groups. So H atoms from \(Ca(OH)_2\): \(2\times1 = 2\).
Step3: Sum the H atoms
Total H atoms on left side: \(8 + 2 = 10\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
10