QUESTION IMAGE
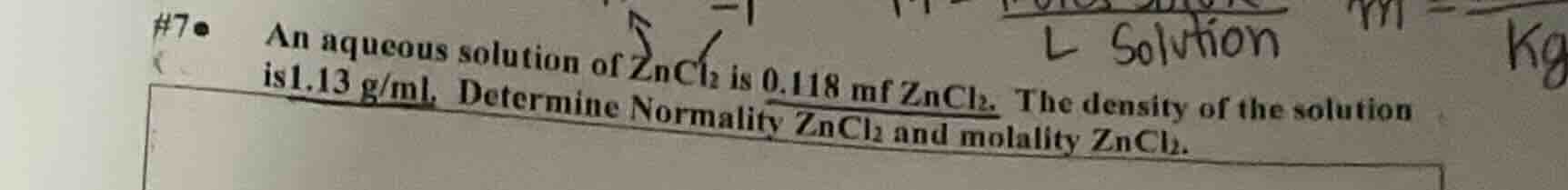
Question
#7 an aqueous solution of zncl₂ is 0.118 mf zncl₂. the density of the solution is 1.13 g/ml. determine normality zncl₂ and molality zncl₂.
Step1: Define given values
Molarity of $\text{ZnCl}_2$ $M = 0.118\ \text{mol/L}$, density of solution $
ho = 1.13\ \text{g/mL} = 1130\ \text{g/L}$, molar mass of $\text{ZnCl}_2$ $MM = 65.38 + 2\times35.45 = 136.28\ \text{g/mol}$
Step2: Calculate Normality
Normality = Molarity × total positive charge. $\text{Zn}^{2+}$ has +2 charge, so:
$\text{Normality} = 0.118 \times 2 = 0.236$
Step3: Find mass of 1L solution
Mass of solution = density × volume:
$m_{\text{solution}} = 1130\ \text{g/L} \times 1\ \text{L} = 1130\ \text{g}$
Step4: Find mass of solute
Mass of $\text{ZnCl}_2$ = moles × molar mass:
$m_{\text{solute}} = 0.118\ \text{mol} \times 136.28\ \text{g/mol} = 16.081\ \text{g}$
Step5: Calculate mass of solvent
Mass of solvent = mass of solution - mass of solute:
$m_{\text{solvent}} = 1130\ \text{g} - 16.081\ \text{g} = 1113.919\ \text{g} = 1.1139\ \text{kg}$
Step6: Calculate Molality
Molality = moles of solute / kg of solvent:
$\text{Molality} = \frac{0.118\ \text{mol}}{1.1139\ \text{kg}} \approx 0.106$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Normality of $\text{ZnCl}_2$: $0.236\ \text{N}$
Molality of $\text{ZnCl}_2$: $0.106\ \text{m}$