QUESTION IMAGE
Question
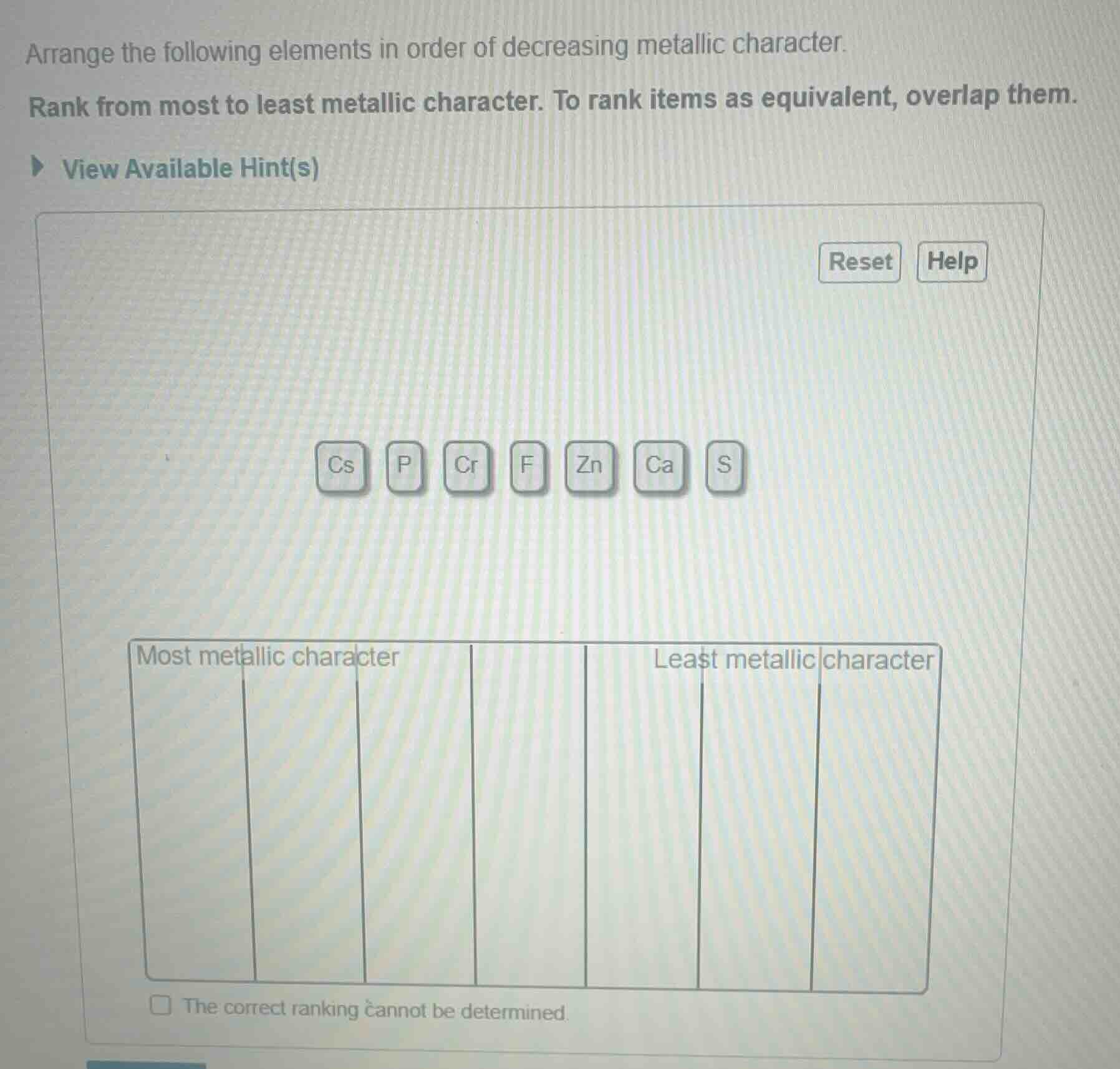
arrange the following elements in order of decreasing metallic character. rank from most to least metallic character. to rank items as equivalent, overlap them. view available hint(s) reset help cs p cr f zn ca s most metallic character least metallic character the correct ranking cannot be determined.
Metallic character increases down a group and decreases across a period (left to right) on the periodic table. Cesium (Cs) is an alkali metal in the lowest period, so it has the highest metallic character. Calcium (Ca) is an alkaline earth metal, followed by transition metals Chromium (Cr) and Zinc (Zn). Nonmetals Phosphorus (P), Sulfur (S), and Fluorine (F) have lower metallic character, with F (a halogen, top right of the table) having the least.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Most metallic character → Cs, Ca, Cr, Zn, P, S, F → Least metallic character