QUESTION IMAGE
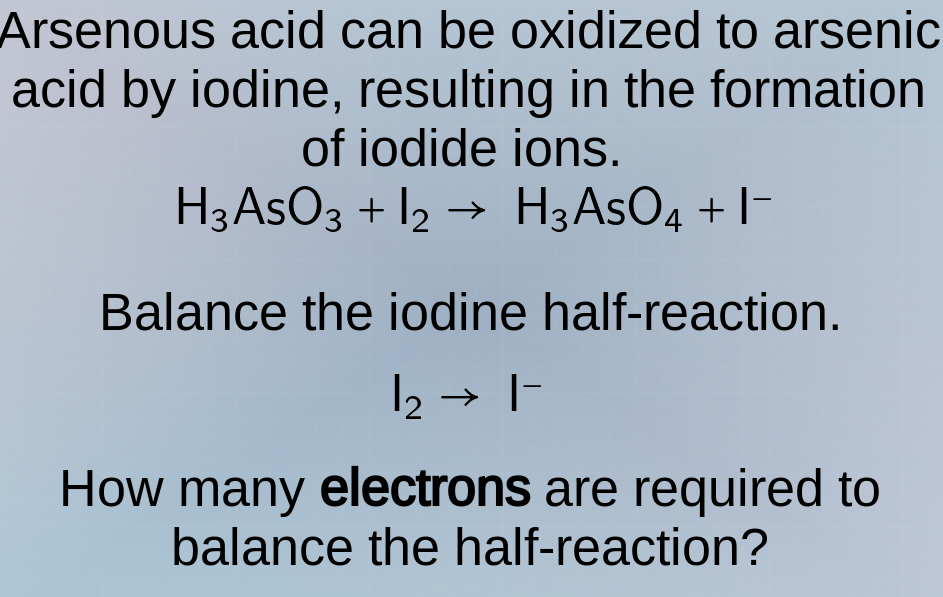
Question
arsenous acid can be oxidized to arsenic acid by iodine, resulting in the formation of iodide ions.
h₃aso₃ + i₂ → h₃aso₄ + i⁻
balance the iodine half - reaction.
i₂ → i⁻
how many electrons are required to balance the half - reaction?
Step1: Balance atoms
First, balance the iodine atoms. There are 2 I atoms in \( I_2 \) and 1 I atom in \( I^- \), so we multiply \( I^- \) by 2:
\( I_2
ightarrow 2I^- \)
Step2: Balance charge
The charge on the left side ( \( I_2 \) ) is 0. The charge on the right side ( \( 2I^- \) ) is \( 2\times(-1) = -2 \). To balance the charge, we need to add 2 electrons to the left side (since electrons have a negative charge, adding electrons to the left will decrease the positive charge or increase the negative charge to match the right side). The half - reaction becomes:
\( I_2 + 2e^-
ightarrow 2I^- \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
2