QUESTION IMAGE
Question
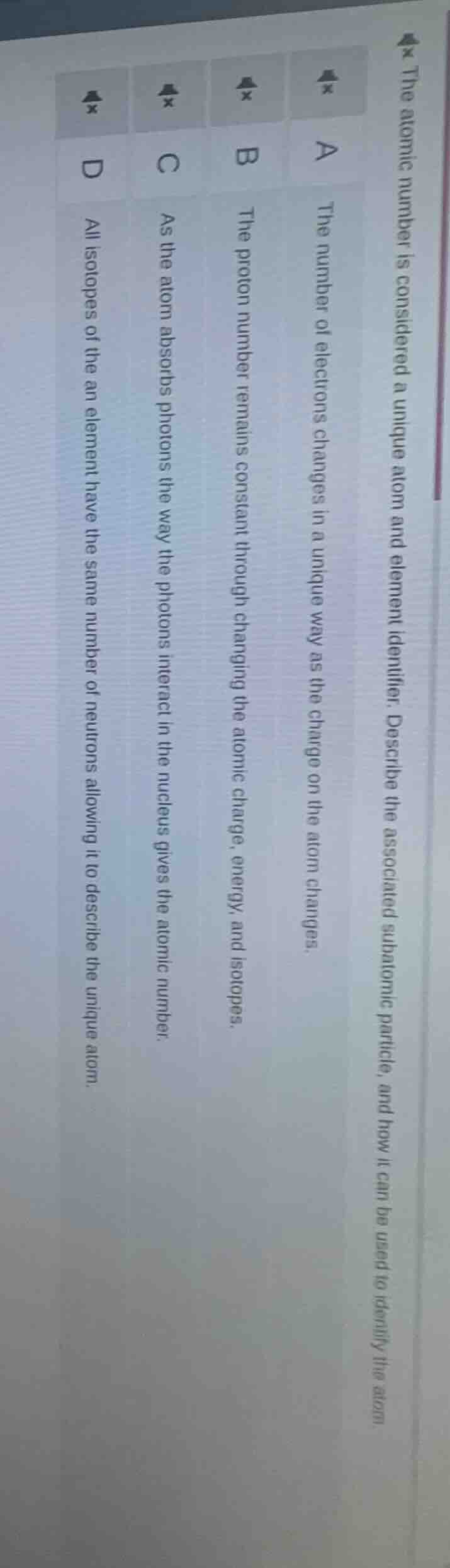
the atomic number is considered a unique atom and element identifier. describe the associated subatomic particle, and how it can be used to identify the atom.
a the number of electrons changes in a unique way as the charge on the atom changes.
b the proton number remains constant through changing the atomic charge, energy, and isotopes.
c as the atom absorbs photons the way the photons interact in the nucleus gives the atomic number.
d all isotopes of the an element have the same number of neutrons allowing it to describe the unique atom.
To determine the correct option, we analyze each choice:
- Option A: The number of electrons can change (e.g., in ions), but this doesn't define the atomic number (which is protons). So A is incorrect.
- Option B: The atomic number is the number of protons. Protons remain constant in an element (even with charge changes, isotopes, etc.). This matches the definition of atomic number as a unique identifier (same element has same proton number).
- Option C: Atomic number is about protons, not photon absorption. So C is incorrect.
- Option D: Isotopes have different neutrons, same protons (atomic number). So D is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The proton number remains constant through changing the atomic charge, energy, and isotopes.