QUESTION IMAGE
Question
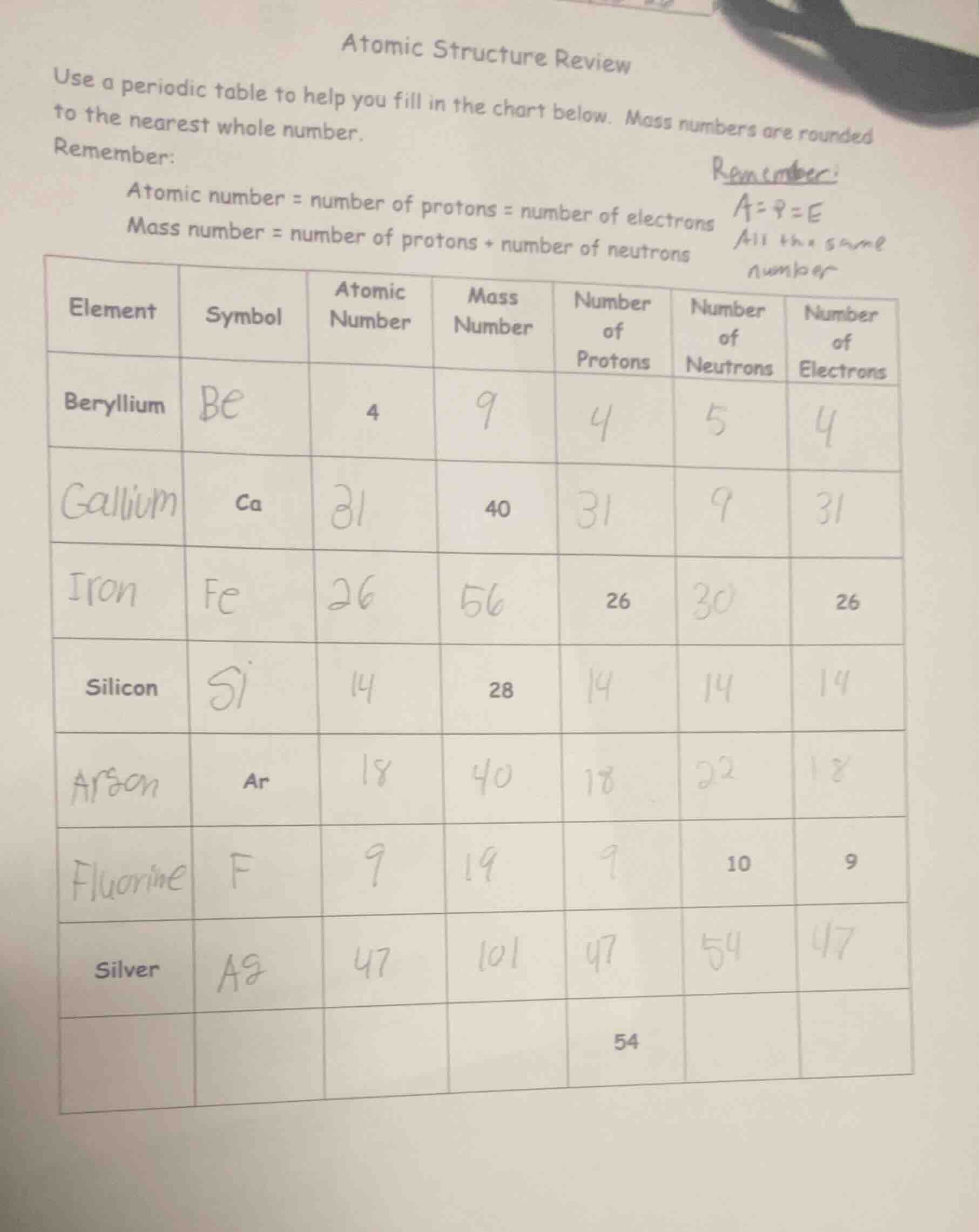
atomic structure review
use a periodic table to help you fill in the chart below. mass numbers are rounded to the nearest whole number.
remember:
atomic number = number of protons = number of electrons
mass number = number of protons + number of neutrons
(the chart part is omitted as per requirements, with columns: element, symbol, atomic number, mass number, number of protons, number of neutrons, number of electrons, and rows for beryllium, gallium, iron, silicon, (a misspelled) arson (should be argon?), fluorine, silver, and a blank row with 54 in protons column)
To solve the atomic structure problems (correcting errors and filling the last row), we use the rules:
- Atomic number (\(Z\)) = protons (\(p^+\)) = electrons (\(e^-\))
- Mass number (\(A\)) = \(p^+ +\) neutrons (\(n^0\)) → \(n^0 = A - p^+\)
Step 1: Identify Errors in Given Table
- Gallium: Symbol should be \( \text{Ga} \) (not \( \text{Ca} \)), Mass number for Ga is ~69.72 (rounded to 70), so \( n^0 = 70 - 31 = 39 \) (not 9).
- Argon: Element name is "Argon" (not "Arson").
- Last Row (Xenon, since \( p^+ = 54 \)):
- Atomic number \( Z = 54 \) (matches \( p^+ = 54 \)).
- Mass number for Xenon (Xe) is ~131.29 (rounded to 131), so \( n^0 = 131 - 54 = 77 \), \( e^- = 54 \).
Step 2: Correct/Fill the Table (Key Rows)
| Element | Symbol | Atomic Number | Mass Number | Protons | Neutrons | Electrons |
|---|---|---|---|---|---|---|
| Argon | \( \text{Ar} \) | 18 | 40 | 18 | 22 | 18 |
| Xenon (Last Row) | \( \text{Xe} \) | 54 | 131 | 54 | 77 | 54 |
Final Corrected Table (Key Fixes)
- Gallium: Symbol \( \text{Ga} \), Mass # 70, Neutrons \( 70 - 31 = 39 \).
- Argon: Element name "Argon".
- Last Row: Element = Xenon (\( \text{Xe} \)), Atomic # 54, Mass # 131, Neutrons \( 131 - 54 = 77 \), Electrons 54.
(Note: For the last row, since protons = 54, the element is Xenon (\( \text{Xe} \)) from the periodic table.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Key Corrections)
- Gallium symbol: \( \boldsymbol{\text{Ga}} \), Mass # \( \boldsymbol{70} \), Neutrons \( \boldsymbol{39} \).
- Argon: Element name \( \boldsymbol{\text{Argon}} \).
- Last row (Xenon): Element \( \boldsymbol{\text{Xenon (Xe)}} \), Mass # \( \boldsymbol{131} \), Neutrons \( \boldsymbol{77} \), Electrons \( \boldsymbol{54} \).