QUESTION IMAGE
Question
2.3 atomic structure and symbolism
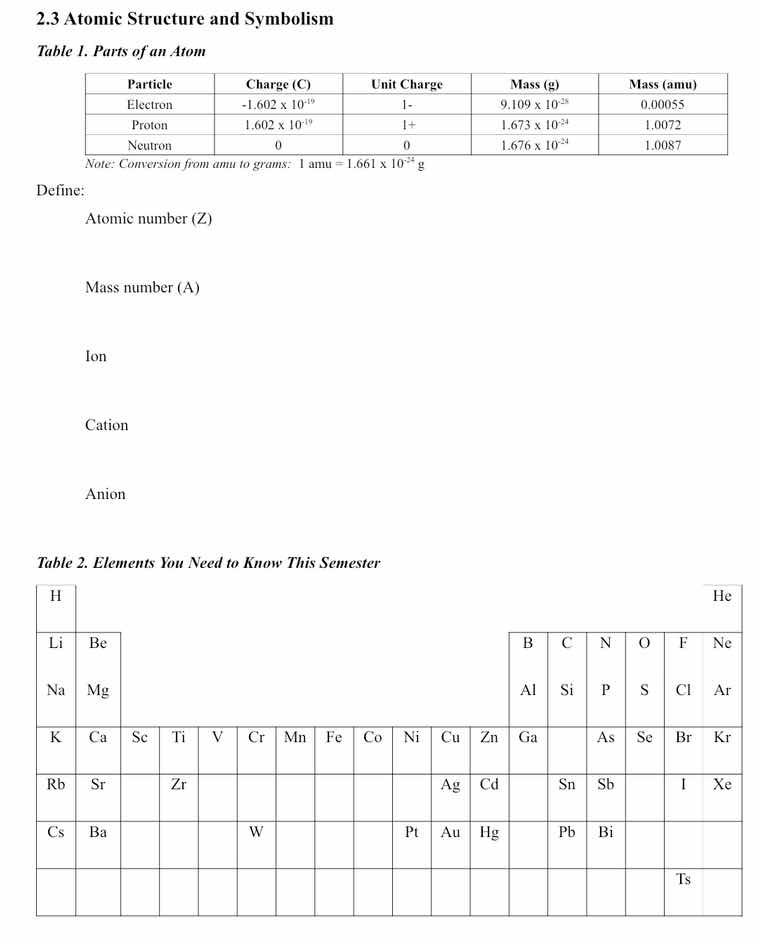
table 1. parts of an atom
| particle | charge (c) | unit charge | mass (g) | mass (amu) |
|---|---|---|---|---|
| proton | 1.602 × 10⁻¹⁹ | 1+ | 1.673 × 10⁻²⁴ | 1.0072 |
| neutron | 0 | 0 | 1.676 × 10⁻²⁴ | 1.0087 |
note: conversion from amu to grams: 1 amu = 1.661 × 10⁻²⁴ g
define:
atomic number (z)
mass number (a)
ion
cation
anion
table 2. elements you need to know this semester
| li | be | b | c | n | o | f | ne | ||||||||||
| na | mg | al | si | p | s | cl | ar | ||||||||||
| k | ca | sc | ti | v | cr | mn | fe | co | ni | cu | zn | ga | as | se | br | kr | |
| rb | sr | zr | sn | sb | i | xe | |||||||||||
| cs | ba | w | pt | au | hg | pb | bi | ||||||||||
| ts |
Brief Explanations
- Atomic number (Z): It is a unique identifier for each element, equal to the number of protons in the nucleus of an atom of that element. In a neutral atom, it also matches the number of electrons.
- Mass number (A): This is the total number of protons and neutrons (collectively called nucleons) in the nucleus of a single atom. It is a whole number approximation of an atom's mass.
- Ion: An atom or molecule that has gained or lost one or more valence electrons, resulting in a net positive or negative electrical charge (no longer electrically neutral).
- Cation: A positively charged ion formed when an atom or molecule loses one or more valence electrons, leaving more protons than electrons.
- Anion: A negatively charged ion formed when an atom or molecule gains one or more valence electrons, leaving more electrons than protons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Atomic number (Z): The number of protons in an atom's nucleus; defines an element.
- Mass number (A): The total number of protons + neutrons in an atom's nucleus.
- Ion: A charged atom/molecule from electron gain/loss.
- Cation: Positively charged ion from electron loss.
- Anion: Negatively charged ion from electron gain.